Group a used 5.00 moles of styrene and 0.0010 moles of benzoyl peroxide while group b used the same amount of benzoyl peroxide as group a but used only 1.50 moles styrene. what would the average molecular weight of each group's polymer sample be
Solved
Show answers
More tips
- F Family and Home How to Quickly and Effectively Remove Gum from Clothing?...
- A Auto and Moto How to choose the right drive for your BMW...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
Answers on questions: Chemistry
- C Chemistry Answer of this question...
- C Chemistry Which statement is FALSE about Dalton s atomic model? Each element has its own kind of atom. Compounds are created when atoms of different elements combine. Atoms cannot...
- C Chemistry What conclusions can you make about the relationship between the volume of a gas and its pressure?...
- C Chemistry An equilibrium mixture contains 0.600 mol of each of the products (carbon dioxide and hydrogen gas) and 0.200 mol of each of the reactants (carbon monoxide and water...
- C Chemistry If a bottle of olive oil contains 1.2 kg of olive oil, what is the volume, in milliliters (mL), of the olive oil?...
- C Chemistry Name the compound Ga S3...
- C Chemistry How did the change of stress (adding or removing reactants or products) cause a shift in the equilibrium system of your solutions? Use data to support your answer. Make...
- C Chemistry Looking at the car in the picture above, explain how the electrons act to form this compound that is on the surface of the car, and what type of ions each element becomes....
- B Business Anegative relationship between the quantity demanded and price is called the law of a. efficiency b. supply c. marginality d. demand...
- M Mathematics Emma has 4 1/2 pounds of birdseed. she wants to divide it evenly among 3 birds feeders. how much birdseed should she put in each?...

Ответ:
Answer : Average molecular weight of group A = 104.0276 g
Average molecular weight of group B = 104.0920 g
Solution : Given, Moles of group A styrene = 5 moles
Moles of group A benzoyl peroxide = 0.0010 moles
Moles of group B styrene = 1.5 moles
Moles of group B benzoyl peroxide = 0.0010 moles
Molecular mass of styrene = 104 g/mole
Molecular mass of benzoyl peroxide = 242.23 g/mol
Formula used : Average molar mass = Total mass / Total moles
For group A,
Average molar mass of group A =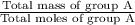
=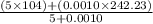
=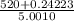
=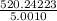
= 104.0276 g
Average molar mass of group A = 104.0276 g
For group B,
Average molar mass of group B =
=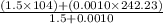
=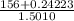
=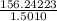
= 104.0920 g
Average molar mass of group B = 104.0920 g
Ответ:
10 moles
Explanation:
set it up as a proportion
450 kL/20 moles = 225 kL/ x moles
Cross multiply to solve for x.