aliveajones2005
03.07.2021 •
Chemistry


GUYS PLSS BEG YOU
can you name all the drawings

Solved
Show answers
More tips
- H Health and Medicine Coughing: Causes, Types, and Treatment Methods...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- H Health and Medicine How to Calm Your Nerves? Expert Tips That Actually Work...
- A Animals and plants 5 Tips for Taking Care of Yews to Keep Them Green and Beautiful...
- S Sport How to wrap boxing hand wraps? Everything you need to know!...
- F Food and Cooking 10 Reasons Why You Should Avoid Giving Re-Gifts: An Informative Guide...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Style and Beauty How Are Eyelash Extensions Applied? All Your Questions Answered...
- F Food and Cooking 10 Tips for Proper Sushi Consumption...
Answers on questions: Chemistry
- M Mathematics Iknow what the domain is, but can someone me find the correct range? ?...
- M Mathematics If a taxpayer does not have enough taxes withdrawn from her paychecks throughout the year to equal the total tax she is required to pay that year, she will owe the government the...
- B Biology Humans have chromosome pairs. DONE...
- M Mathematics I need help with all the math please all of it...
- G Geography The language and religion of france developed from which people? a. the franks b. the gauls c. the anglo-saxons d. the romans...

Ответ:
Explanation:
Jejwuiwiwiw88w83747
Ответ:
Answer : 20.1 grams
Explanation:
According to avogadro's law, 1 mole of every substance occupies 22.4 L at STP and contains avogadro's number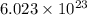 of particles.
of particles.
To calculate the number of moles, we use the equation:
For nitrogen:
For hydrogen gas:
The chemical equation for the reaction is:
By Stoichiometry of the reaction:
1 mole of nitrogen gas reacts with 3 moles of hydrogen.
So, 0.591 moles of nitrogen gas will react with = of hydrogen.
of hydrogen.
As, given amount of hydrogen is more than the required amount. So, it is considered as an excess reagent.
Thus, nitrogen gas is considered as a limiting reagent because it limits the formation of product.
By Stoichiometry of the reaction:
1 mole of nitrogen gas produces 2 moles of ammonia
So, 0.591 moles of nitrogen gas will produce =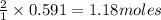 of ammonia
of ammonia
Now, calculating the mass of ammonia t:
Thus theoretical yield of ammonia is 20.1 grams