AjTruu2880
21.11.2019 •
Chemistry
Heat is transferred at a rate of 2 kw from a hot reservoir at 775 k to a cold reservoir at 300 k. calculate the rate at which the entropy of the two reservoirs changes. (round the final answer to six decimal places.)
Solved
Show answers
More tips
- B Business and Finance How to Get Your Money Back When Lending? Top 7 Ways...
- S Sport How to Learn Swimming? Simple Tips for Beginners...
- P Photography and Videography Understanding HDR: How It Works and Why You Need It...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
Answers on questions: Chemistry
- C Chemistry What is between the cornea and iris...
- C Chemistry What do members of the same group have in common?...
- C Chemistry Advantages and disadvantages involved in methods of reproduction for hammerhead hurry...
- B Business 06:02 MC) An automobile company is building a new factory in your town. What may be a positive externality of this event? (1 point) a Noise produced by the workers and factory machines...
- M Mathematics What are the solutions to the system of linear equations graphed below?...
- H History How might the front page help sell the newspaper? ...
- M Mathematics What is the slope of y = 4x + 7?...
- M Mathematics Some one pls help me this work is due in 39 minutes pls help me Write two interges with different signs that have a sum of -25. Write two intergers with the same sign that have a sum...
- M Mathematics Please help As Soon As Possible...
- M Mathematics Solve the following question...

Ответ:
Explanation:
The given data is as follows.
Q = 2 kW,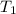 = 775 K
= 775 K
The relation between entropy and heat energy is as follows.
Therefore, calculate the entropy at each temperature as follows.
=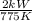
=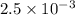 kW/K
kW/K
Also,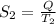
=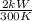
=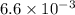 kW/K
kW/K
Hence, the change in entropy will be calculated as follows.
= kW/K
kW/K
=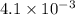 kW/K
kW/K
or, = 0.0041 kW/K
Thus, we can conclude that the rate at which the entropy of the two reservoirs changes is 0.0041 kW/K.
Ответ:
Thank you very much~ Ciel Phantomhive