justinpatengale2002
25.09.2019 •
Chemistry
Heat is transferred by conduction through a wall of thickness 0.87 m. what is the rate of heat transfer if the walls thermal conductivity is 13 w/m-k, the surface area is 5 m2, and the surface temperatures are 14 °c and 93 °c. report your answer in watts.
Solved
Show answers
More tips
- H Health and Medicine Flu: How to Recognize It by the First Symptoms?...
- F Food and Cooking How to Sober Up Quickly? Important Facts and Tips...
- H Health and Medicine How to Properly Take a Blood Sugar Test?...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
Answers on questions: Chemistry
- M Mathematics (Please answer this asap) Mia has a circular garden that she wants to put a fence around. She knows that the area of a garden is 9p yds^2. How much fencing does Mia need to go around...
- M Mathematics A scale says that it s readings are accurate to within 1%, meaning that when an object is weighed using that scale, the reading will have an error of at most 1%. Gillian puts a math...
- E English How did Dally get discharged from the hospital so quickly?(The outsiders chapter 9)...

Ответ:
Explanation:
The given data is as follows.
Thickness (dx) = 0.87 m, thermal conductivity (k) = 13 W/m-K
Surface area (A) = 5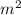 ,
, 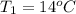
According to Fourier's law,
Q =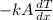
Hence, putting the given values into the above formula as follows.
Q =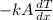
=
= 5902.298 W
Therefore, we can conclude that the rate of heat transfer is 5902.298 W.
Ответ:
Hello!
1.00 L of a gas at STP is compressed to 473 mL. What is the new pressure of gas?
We have the following data:Vo (initial volume) = 1.00 L
V (final volume) = 473 mL → 0.473 L
Po (initial pressure) = 1 atm (pressure exerted by the atmosphere - in STP)
P (final pressure) = ? (in atm)
We have an isothermal transformation, that is, its temperature remains constant, if the volume of the gas in the container decreases, so its pressure increases. Applying the data to the equation Boyle-Mariotte, we have:The new pressure of the gas is 2.11 atm
___________________________________