trapvannah
17.12.2020 •
Chemistry
Helpppppppppppppppppppppp
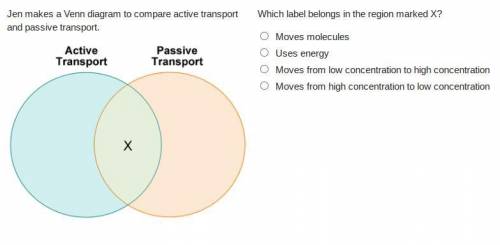
Solved
Show answers
More tips
- P Philosophy How did the concept of module arise in computer science?...
- D Dating, Love, Relationships Why Should the Man be Active and the Woman Passive during Foreplay?...
- S Society and Politics Выборы: Смысл, Значение и Отражение...
- B Business and Finance How to Get Your Money Back When Lending? Top 7 Ways...
- F Family and Home Do Lullabies Help Babies Sleep or Is it Just a Myth?...
- F Family and Home Why Does God Punish Us Constantly and How Can We Fix It?...
- D Dating, Love, Relationships Why do we feel shame?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- O Other How to Accidentally Get a Rare Coin with Your Change and How to Know Its Value?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
Answers on questions: Chemistry
- G Geography True or false mid-ocean ridges form the longest mountain range on earth...
- M Mathematics A company says its premium mixture of nuts contains % Brazil nuts, % cashews, % almonds, and % hazelnuts, and the rest are peanuts. You buy a large can and separate...
- E English Most of the events in the play are presented in flashback flashforward diary entries a monologue...
- M Mathematics 1point 8. four negatively charged particles are at various distances from a positively charged plate. the partides are equally charged and do not influence each other....
- E English Help me with this please...

Ответ:
Ответ:
Ответ:
Assume 100 g of unknown compound.
50.0⋅g 12.011⋅g⋅mol−1 = 4.163 mol⋅C.
6.7⋅g 1.00794⋅g⋅mol−1 = 6.65 mol⋅H.
43.3⋅g 15.999⋅g⋅mol−1 = 2.71 mol⋅O.
divide thru by the lowest molar quantity, that of oxygen to get a formula of C1.54H2.45O1, double to give a whole number EMPIRICAL formula of C3H5O2