jacksoneaton
02.10.2021 •
Chemistry
How long does it take for carbon monoxide to dissipate?
Solved
Show answers
More tips
- H Health and Medicine How to Get Pregnant Faster?...
- S Style and Beauty Lamination of Hair: How it Works and What it is?...
- C Computers and Internet Best Applications for Your iPad: Review of the Best Candidates for Installation...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
Answers on questions: Chemistry
- C Chemistry rections. Correctly . Molecules of a substance are in motion (only as a gas, only above the freezing point, all of the time). lectrical) onero roy of the molecules,...
- C Chemistry Why is the angle of the orbit the moon uses to revolve around the sun...
- H History Factors that contributed to yemen becoming a failed state. social studies. and if you have the answers for this you give them to...
- M Mathematics Translate the following written expression into a mathematical expression. Then, find the expression s value when a = 234 and b = 4. The difference of the quotient of a and 6 and...
- M Mathematics The given sphere has a radius of 2 inches. What happens to the volume if the radius is doubled?...

Ответ:
The half-life of carboxyhemoglobin in fresh air is approximately 4 hours. To completely flush the carbon monoxide from the body requires several hours, valuable time when additional damage can occur.
Symptoms: Dizziness; Unconsciousness; Confusion...
Explanation:
Ответ:
Explanation:
According to the mole concept, there are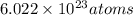 present in 1 mole of a substance.
present in 1 mole of a substance.
Mathematically, 1 mole =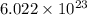 atoms or molecules
atoms or molecules
Therefore, calculate the number of moles of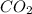 present in
present in 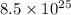 molecules as follows.
molecules as follows.
= mol
mol
or, = 141.1 mol
Thus, we can conclude that there are 141.1 moles present in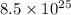 molecules of
molecules of 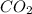 .
.