alligatorsocks
05.10.2019 •
Chemistry
How many calcium ions are there in caco3?
Solved
Show answers
More tips
- L Leisure and Entertainment When will Maslenitsa start?...
- F Food and Cooking Discovering the Mysterious Fruit of Feijoa...
- B Business and Finance How to Open an Online Store? A Detailed Guide for Beginners...
- W Work and Career How to Write a Resume That Catches the Employer s Attention?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
Answers on questions: Chemistry
- C Chemistry The equation below shows the formation of ammonia. If 56g of nitrogen gas is used in the reaction, how much ammonia will be formed? Use the table to help you. Enter...
- C Chemistry A 5cm string that is fastened at both sides is plucked. What is the wavelength of the n = 2 standing wave?...
- C Chemistry Chlorination of alkanes can produce a multitude of products. determine the number of monochlorinated and dichlorinated products that can be obtained by chlorination...
- C Chemistry Consider a different analyte for this exercise. citric acid is found in many fruits and fruit juices. sodium hydroxide (naoh) is the titrant and citric acid (h3c6h5o7)...
- C Chemistry You determine the volume of your plastic bag (simulated human stomach) is 1.08 l. how many grams of nahco3 (s) are required to fill this container given a 49.4% co2...
- C Chemistry How many mL can you make of a .225 M solution if you have 458 mL of a .500 M solution available?...
- C Chemistry Solid iron metal reacts with solid sulfur at room temperature to produce solid iron (ll) sulfide? Express as a chemical equation....
- C Chemistry A block is pulled 0.90 m to the right in 4.5 s. What is the block s average speed to the nearest tenth of m/s?...
- M Mathematics What property is -2x–6+4=-2? (see image)...
- W World Languages Kon chodna chahta hai?...

Ответ:
The density of the metal object=6.0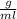
Given:
Volume of the metal object=1.5ml
Mass of the metal object=9.0g
To find:
Density of the metal object
Step by Step Explanation:
Solution:
According to the formula, Density of the metal object can be calculated as
Where, m=mass of the metal object
v=volume of the metal object
We know the values of v=1.5ml and m=9.0g
Substitute these values in the above equation we get
=6.0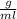
Result:
Thus the density of the metal object is 6.0