pleasehelp5334me2
28.06.2019 •
Chemistry
How many grams of carbon monoxide are needed to react with an excess of iron (iii) oxide to produce 198.5 grams of iron? fe2o3(s) + 3co(g) à 3co2(g) + 2fe(s)
Solved
Show answers
More tips
- A Animals and plants How to Grow Lime from a Seed: Simple Tips and Interesting Facts...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
Answers on questions: Chemistry
- C Chemistry The volume of a sample of chlorine gas is 8.00 liters at 45.0 °C and 0.966 atm. how many moles of chlorine are present in the sample? r=0.0821...
- C Chemistry Four coordinate nickel complexes can have another geometry other than tetrahedral. Name that geometry, and explain why the absorption spectrum you took strongly suggests...
- C Chemistry Lead nitrate undergoes thermal decomposition according the following reaction.2Pb(NO3)2 (s) ⟶ 2PbO (s) + 4NO2 (g) + O2 (g) ΔH = 608 kJWhat is the ΔH for the reaction:6Pb(NO3)2...
- C Chemistry Draw 7. Draw a yellow arrow to indicate the incoming visible light. Draw red arrows to indicate the paths of infrared energy. Label arrows accordingly. Earth s surface...
- C Chemistry Mi amiga necesita novia y ¿hay alguna chica que quiera salir con ella?...
- C Chemistry Write chemical equations for the following chemical reactions: a)decomposition of zinc carbonateb)decomposition of lead nitratec(decomposition of ammonium dichromated)Reaction...
- S Social Studies Please help me with this 5 questions!...
- M Mathematics Can someone help with this one...
- A Arts I like ya cut g :)))))...
- E English Think about the author s use of structure in your Module One short story. How does the author order events? What plot techniques (flashback, flash forward, dream sequence)...

Ответ:
Answer : The grams of carbon monoxide needed are 148.89 g
Solution : Given,
Mass of iron, Fe = 198.5 g
Molar mass of iron, Fe = 56 g/mole
Molar mass of carbon monoxide, CO = 28 g/mole
First we have to calculate the moles of iron, Fe.
The balanced chemical reaction is,
From the balanced reaction, we conclude that
2 moles of iron produces from the 3 moles of carbon monoxide
3.545 moles of iron produces from the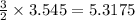 moles of carbon monoxide
moles of carbon monoxide
Now we have to calculate the mass of carbon monoxide, CO.
Therefore, the grams of carbon monoxide needed are 148.89 g
Ответ:
One way the modern atomic model differs from Thomson's atomic model is that electrons are no longer scattered throughout an atom's positive matter like chocolate chips in a cookie dough. instead, each electron is outside of the nucleus occupying a specific Energy Level.
The modern atomic model is also called the Schrodinger's model or the Quantum Mechanical model. Although atoms are commonly depicted like planets with electrons orbiting it like satellites, in reality, electrons aren't in fixed orbits but there are clouds where there is a bigger probability of finding an electron. This area is called an Energy Level.
Have a nice day!