dantelin123
11.04.2020 •
Chemistry
How many grams of CO₂ will be produced from 12.0 g of CH₄ and 133 g of O₂?
Solved
Show answers
More tips
- P Philosophy How did the concept of module arise in computer science?...
- D Dating, Love, Relationships Why Should the Man be Active and the Woman Passive during Foreplay?...
- S Society and Politics Выборы: Смысл, Значение и Отражение...
- B Business and Finance How to Get Your Money Back When Lending? Top 7 Ways...
- F Family and Home Do Lullabies Help Babies Sleep or Is it Just a Myth?...
- F Family and Home Why Does God Punish Us Constantly and How Can We Fix It?...
- D Dating, Love, Relationships Why do we feel shame?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- O Other How to Accidentally Get a Rare Coin with Your Change and How to Know Its Value?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
Answers on questions: Chemistry
- C Chemistry Who might benefit financially the most from the epa s claim...
- C Chemistry Why blue intensity of copper(2)sulphate will decrease in a voltaic cell with zinc electrode in zinc sulphate solution and copper 2sulphate solution with copper electrode...
- C Chemistry Map university science books presented by sapling leaming eral chemistry 4th edition balance the following equation in acidic conditions. phases are optional. tip: if...
- C Chemistry How much work is done by a 35-watt light bulb if it is on for 1,320 seconds? plz help i need to do good on this test...
- C Chemistry Describe the difference in pressure against your cheeks when they were full of air versus when you released it. how were you able to release the air? in which direction...
- C Chemistry How many grams of zno would you need to produce 50 g of zncl2? (the atomic mass of zn is 65.38 and of cl is 35.45.) zno(s) + 2hcl(aq) → zncl2 (s) + h2o(l) 17.1 g 29.8...
- E English How did the Phoenicians turn imports from other cultures into exports?...
- M Mathematics PLS HELP I AM BEING TIMEDD...
- B Business Im sorry that i left but i just missed everyone. Especially Alphonso, because he was the nicest person i ever met, literally. Just know i miss all of my friends, and...
- P Physics Zoom anyone? (dont report) meeting id- 216-907-4596/ passcode is ath2TM...

Ответ:
mass of CO_2 formed=
Explanation:
First balance the chemical reaction:
molecular mass of
molecular mass of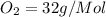
mass given of
mole of
mass given of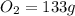
mole of
from the above balanced equation:
1 mole of need 2 mole of
need 2 mole of  gas
gas
therefore 0.75 mole of will react with 1.5 mole of
will react with 1.5 mole of  only
only
but we have 4.16 mole gas
gas
hence will be the excess reagent and
will be the excess reagent and  will be the limiting eagent so production of
will be the limiting eagent so production of  will depenf on the quantity of limiing reagent i.e.
will depenf on the quantity of limiing reagent i.e. 
from the above balanced equation:
1 mole of gives 1 mole of
gives 1 mole of  on complete reacion with
on complete reacion with 
hence,
0.75 mole of gives 0.75 mole of
gives 0.75 mole of  on complete reacion with
on complete reacion with 
mass of CO_2 formed=
Ответ:
just guess
Explanation:
if you do not like the answer that i have presented you with on this fine day then you are a smelly sea pickle.