dashaefairley2
16.09.2019 •
Chemistry
How many grams of oxygen are needed to react completely with 200.0 g of ammonia, nh3? 4nh3(g) + 5o2(g) ? 4no(g) + 6h2o(g)?
Solved
Show answers
More tips
Answers on questions: Chemistry
- C Chemistry Ill give u brainliest and all my points! pls explain using complete sentences where on the hr diagram you would expect to find a main sequence star based on its physical...
- C Chemistry Is carbon monoxide is a poisonous gas....
- C Chemistry Score: ammonium hydroxide ionizes as follows: nh4oh nh4+ + oh-. if an excess of ammonium salt, such as nh4cl (which ionizes to give nh4+ ions), is added to the equilibrium...
- C Chemistry The overall charge of an ionic compound is a. always positive b. usually positive c. usually negative d. usually neutral...
- C Chemistry Nitrogen in air reacts at high temperature to form no2 according to the reaction: n2 + 2 o2 ? 2 no2 draw structures for the reactants and products indicating the number...
- C Chemistry A solution of ammonia NH3(aq) is at equilibrium. How would the equilibrium change if OH were added to the solution? A. (NH4+) and (OH) would decrease. O B. (NH4+] would...
- C Chemistry PLEASE HELP!!! Explain how an electric car might still run on fossil fuels....
- E English Point of View Task What is the point of view of the passage? Beep-bo-bo-beep! Tommy s phone beeped as he collected the glowing banana. Do-do-do-dee! Tommy s phone hummed...
- M Mathematics I need my Sons help how do you compare 4/10 and 8/12?...
- M Mathematics If we select a student at random, the marginal probability that they are in the Health Science program, that is, P(student is in Health Science) is approximately Group of...

Ответ:
Answer : The mass of oxygen needed are 470.592 grams.
Solution : Given,
Mass of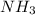 = 200.0 g
= 200.0 g
Molar mass of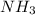 = 17 g/mole
= 17 g/mole
Molar mass of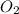 = 32 g/mole
= 32 g/mole
First we have to calculate the moles of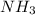 .
.
Now we have to calculate the moles of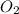
The balanced chemical reaction is,
From the reaction, we conclude that
As, 4 mole of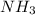 react with 5 mole of
react with 5 mole of 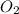
So, 11.765 moles of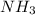 react with
react with 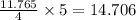 moles of
moles of 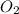
Now we have to calculate the mass of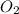
Therefore, the mass of oxygen needed are 470.592 grams.
Ответ:
hi today was normal
Explanation:
nothing great