How many grams of oxygen can be produced from the decomposition of 100. g of kclo3?
Solved
Show answers
More tips
- F Food and Cooking 10 Ideas for a Wedding Anniversary Gift...
- H Health and Medicine Hangover: How to Get Rid of It Quickly?...
- S Style and Beauty How to Choose the Right Fur Coat and Avoid Regrets?...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- C Chemistry The Earth spinning on its axis helps us determine a measurement of time. Which sentence from the article provides the BEST support to the statement above? a (A)...
- M Mathematics Solve for x. this is very confusing...
- M Mathematics Simon makes eleven dollars an hour. A 2-column table with 5 rows. Column 1 is labeled Days with entries 1, 2, 3, 4, 5. Column 2 is labeled miles with entries 11,...
- P Physics Both a gage and a manometer are attached to a gas tank to measure its pressure. if the reading on the pressure gage is 80 kpa, determine the distance between the...

Ответ:
39.168g of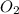
Explanation:
Step 1: Determine the number of moles of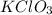
where 'n' is the number of moles, m is the mass of compound and M is the molar mass of compound
Step 2: Determine number of moles of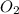 produced per mole of
produced per mole of 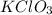 decomposed
decomposed
1 mole of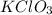 will produce 1.5 moles of
will produce 1.5 moles of 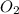
Step 3: Determine mass of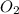
Ответ:
To solve this we use the equation,
M1V1 = M2V2
where M1 is the concentration of the stock solution, V1 is the volume of the stock solution, M2 is the concentration of the new solution and V2 is its volume.
3 M x 2 mL = M2 x 250 mL
M2 = 0.024 M