vickygloom
02.07.2019 •
Chemistry
How many grams of pb are contained in a mixture of 0.135 kg each of pbcl(oh) and pb2cl2co3?
Solved
Show answers
More tips
- C Computers and Internet War of Social Media: Which Platform is the Leader?...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- H Health and Medicine How to Calm Your Nerves? Expert Tips That Actually Work...
- A Animals and plants 5 Tips for Taking Care of Yews to Keep Them Green and Beautiful...
- S Sport How to wrap boxing hand wraps? Everything you need to know!...
- F Food and Cooking 10 Reasons Why You Should Avoid Giving Re-Gifts: An Informative Guide...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Style and Beauty How Are Eyelash Extensions Applied? All Your Questions Answered...
- F Food and Cooking 10 Tips for Proper Sushi Consumption...
Answers on questions: Chemistry
- C Chemistry Ineed with questions 16-21 and it’s very hard and i’m struggling with it and if you need to see the picture big then click on it and it will be big...
- C Chemistry Acompound has an empirical formula of chs. what is true about this empirical formula? it models the structure of the compound.b.it shows the ratio of elements in the...
- M Mathematics Soon after taking an aspirin, a paitent has absorbed 300 milligrams of the drug. If the amount of aspirin in the bloodstream decays exponentially with half being removed...
- E English my parents really created a Frankenstein s monster with my brother is an example of what kind of allusion? A) Historical allusion B) Biblical allusion C) Literary...
- E English Brainiest: Which resource: the video, the photographs, the text or the poem will help you the most complete the performance task about the 1906 Earthquake?...
- S Social Studies Which number represents Lake Victoria? A) 1 B) 2 C) 3 D) 4...

Ответ:
Mass of lead in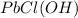 is 107.74 g and mass of lead in
is 107.74 g and mass of lead in 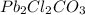 is 103.6 g
is 103.6 g
Explanation:
To calculate the number of moles, we use the equation:
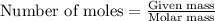 .....(1)
.....(1)
For PbCl(OH):Given mass of PbCl(OH) = 0.135 kg = 135 g (Conversion factor: 1 kg = 1000 g)
Molar mass of PbCl(OH) = 259.66 g/mol
Putting values in equation 1, we get:
In 1 mole of PbCl(OH), 1 mole of lead, 1 mole of chlorine, 1 mole of hydrogen and 1 mole of oxygen atoms are present.
Moles of lead =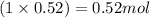
Calculating the mass of lead in PbCl(OH) by using equation 1, we get:
Molar mass of lead atom = 207.2 g/mol
Moles of lead atom = 0.52 moles
Putting values in equation 1, we get:
Mass of lead atom = 107.74 g
ForGiven mass of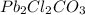 = 0.135 kg = 135 g
= 0.135 kg = 135 g
Molar mass of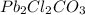 = 545.3 g/mol
= 545.3 g/mol
Putting values in equation 1, we get:
In 1 mole of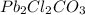 , 2 moles of lead, 2 moles of chlorine, 1 mole of carbon and 3 moles of oxygen atoms are present.
, 2 moles of lead, 2 moles of chlorine, 1 mole of carbon and 3 moles of oxygen atoms are present.
Moles of lead =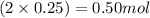
Calculating the mass of lead in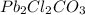 by using equation 1, we get:
by using equation 1, we get:
Molar mass of lead atom = 207.2 g/mol
Moles of lead atom = 0.50 moles
Putting values in equation 1, we get:
Mass of lead atom = 103.6 g
Hence, mass of lead in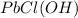 is 107.74 g and mass of lead in
is 107.74 g and mass of lead in 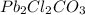 is 103.6 g
is 103.6 g
Ответ:
When 1.20 mole of ammonia reacts, 1.8 moles of water are produced.
Explanation:
The balanced reaction is:
4 NH₃(g) + 5 O₂(g) → 4 NO (g) + 6 H₂O
By stoichiometry of the reaction, the following amounts of moles participate in the reaction:
NH₃: 4 moles O₂: 5 molesNO: 4 molesH₂O: 6 molesThen you can apply the following rule of three: if by stoichiometry 4 moles of ammonia produce 6 moles of water, 1.2 moles of ammonia will produce how many moles of water?
moles of water= 1.8 moles
When 1.20 mole of ammonia reacts, 1.8 moles of water are produced.