How many grams of silver sulfide are formed when 0.280 g of hydrogen sulfide reacts with excess silver and oxygen?
Solved
Show answers
More tips
- C Computers and Internet Best Applications for Your iPad: Review of the Best Candidates for Installation...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- L Leisure and Entertainment Carving: History and Techniques for Creating Vegetable and Fruit Decorations...
Answers on questions: Chemistry
- C Chemistry Mercury and bromine will react with each other to produce mercury(II)bromide. What is the sum of the coefficients in the balanced reaction?...
- C Chemistry What makes sound waves different from light waves?...
- C Chemistry True/False –Similar to liquids, gases cannot be compressed....
- C Chemistry The radioactive isotope carbon-14 is used for radiocarbon dating. The half-life of carbon-14 is 5.730 x 109 years. A wooden artifact in a museum has a 14 C count that is 3.13 x 10-2...
- C Chemistry Plz help...How many moles of ammonia (NH3) can be produced from 6 moles of nitrogen (N2) reacting? show work and a correct answer. N (9) + 3H2(g) → → 2NH,(9)...
- C Chemistry Why is having a healthy lion population in an African savanna important for maintaining biodiversity in that ecosystem? C If the population of lions decreases, there would be less...
- C Chemistry 5. The chart lists organisms in five different categories living near the Texas Gulf Coast. Organisms Living Near the Texas Gulf Coast Vegetation Mammals Invertebrates Fish Birds •...
- C Chemistry How many grams are in 3.60 x 1024 molecules of CuCl2?...
- C Chemistry The combustion of isooctane fuel (C8H18) has an enthalpy change of −5,460 kJ/mol. How many kilojoules of energy are released when 350.0 grams of isooctane are burned?...
- C Chemistry What would the frequency be of a photon beam that had 4.72 x 10^8 joules of energy. Planck s constant is 6.6262 x 10^-34 joule* seconds...

Ответ:
Explanation:
For this question we have to start with the reaction:
Now, we can balance the reaction, so:
With this in mind, we have to start with the amount of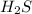 . The first step is to convert from grams to moles. For this, we need to find the molar mass of
. The first step is to convert from grams to moles. For this, we need to find the molar mass of 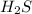 . If we check the periodic table we will find the atomic masses for Ag and H; H: 1 g/mol and A: 32 g/mol, so:
. If we check the periodic table we will find the atomic masses for Ag and H; H: 1 g/mol and A: 32 g/mol, so:
(1*2)+ (32*1) = 34 g/mol.
Now we can calculate the moles of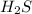 :
:
With the moles of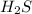 we can calculate the moles of
we can calculate the moles of 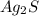 if we check the molar ratio in the balanced equation,
if we check the molar ratio in the balanced equation, 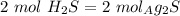 , so:
, so:
With the molar mass of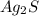 we can convert from moles to grams (Ag: 107.86 g/mol, S: 32 g/mol), so:
we can convert from moles to grams (Ag: 107.86 g/mol, S: 32 g/mol), so:
(107.86*1)+(32*2)=247.80 g/mol
I hope it helps!
Ответ:
I uploaded the answer to a file hosting. Here's link:
cutt.ly/nz6VGda