ayoismeisalex
21.04.2020 •
Chemistry
How many grams of sodium are required to completely react with 19.2 L of Cl, gas at STP according to the following chemical reaction?
Remember 1 mol of an ideal gas has a volume of 22.4 L at STP 2 Na(s) +
Cl2(g) – 2 NaCl(s)
Solved
Show answers
More tips
- O Other What is the oldest joke ever told?...
- F Food and Cooking How to Make Lazy Cabbage Rolls? Simple Steps to a Delicious Dish...
- F Food and Cooking Unusually Delicious Shashlik - Follow the Etiquette of Proper Preparation!...
- L Leisure and Entertainment Couchsurfing: A New Way to Travel...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
Answers on questions: Chemistry
- C Chemistry 3. A 0.600L sample of a 2.50M solution of potassium iodide (KI) contains what mass of KI? Do not include units. Round the the nearest whole number. * (5 Points) Enter...
- C Chemistry A 5.25 L tank holds helium gas at a pressure of 1655 psi. A second 5.25 L tank holds oxygen at a pressure of 440.0 psi. The two gases are mixed in a 5.25 L tank. If the...
- C Chemistry now imagine that you have analyzed an additional vntr. from the blot below, what is the expected frequency of occurrence?...
- C Chemistry Match each element to the correct description of its usual behavior in a chemical reaction. Carbon (C) 12 Will lose electrons Neon (Ne) 2 Will gain electrons Calcium (Ca)...
- C Chemistry Lolies a b c d e f g h i j k l m n o p w r a y y v...
- C Chemistry What does the law say about matter...
- C Chemistry How do the processes of respiration, Combustion, and photosynthesis determine the percentage of carbon dioxide in the atmosphere? a. Photosynthesis…… b. Combustion……...
- C Chemistry Duas espécies , X e Y, pertencentes a uma mesma população ancestral foram isoladas geograficamente por uma barreira, depois de determinado tempo passaram a entrar em contato...
- C Chemistry 12. Chubchub runs a 440m lap in 1.2 minutes. How many laps will Chubchub make if he ran for 50 minutes?...
- C Chemistry Which statement about the manufacture of aluminium by electrolysis is correct? А Aluminium ions are oxidised to aluminium by gaining electrons. B Aluminium is extracted...

Ответ:
12438 grams of sodium are required to completely react with 19.2 L of Cl, gas at STP.
Explanation:
Data given:
mass of sodium required = ?
volume of Chlorine gas = 19.2 L or 19200 grams
Balanced chemical reaction:
2 Na(s) + (g) ⇒ 2 NaCl(s)
(g) ⇒ 2 NaCl(s)
moles of chlorine gas in the reaction is calculated
atomic mass of chlorine gas = 71 grams/litre
number of moles of chlorine gas =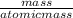
number of moles of chlorine gas =
number of moles = 270.4 moles
from the balanced reaction,
2 moles of Na, and 1 mole of chlorine gas reacts to form 2 moles of NaCl
270.4 moles of chlorine gas is provided so number of moles of Na will be multiplied by 2, using mole ratio
so number of moles of Na = 270.4 x 2
= 540.8 moles
mass of sodium = number of moles x atomic mass
mass of sodium = 540.8 x 23
= 12438 grams
Ответ:
8o/58*6*10power23 gives u the answer. molecules=moles*avagadrono
moles=weight/molecular wt
hope u understandmark me as branliest