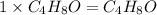sanders8151
18.03.2021 •
Chemistry
How many ions are found in 7.39 g of methanol?
Solved
Show answers
More tips
- S Science and Technology How to Check a Barcode: Unique Product Identifier...
- C Computers and Internet Thin Client: What It Is and Why You Need It?...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- C Computers and Internet Clearing Cache: How to Speed Up Your Browser...
- S Style and Beauty How are artificial nails removed?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- F Food and Cooking Everything You Need to Know About Pasta...
Answers on questions: Chemistry
- C Chemistry Calculate mass in grams of 5.09×10^20 atoms of copper. (Atomic Mass 63.5)...
- C Chemistry Which letter represent a molecule with stronger covalent bonds?...
- C Chemistry What is the chemical equation for the complete combustion of the alkane C2H6 ?...
- C Chemistry Calculate the number of atoms of Barium if the moles are 6.5 mol....
- C Chemistry CHo các nguyên tử X, Y, Z thỏa mãn những điều kiện sau: Nguyên tử X có khối lượng =40, hạt nhân có chứa 22 hạt proton Nguyên tử X có 3 lớp e, lớp ngoài cùng có 7 electron...
- C Chemistry To conclude through reasoning from something known or assumed is to...
- B Business The quick ratio of a firm with current assets of $300,000, current liabilities of $100,000 and inventory of $100,000 is...
- B Business Five years ago, a customer purchased 1,000 shares of ABC stock at $60 per share. The stock has appreciated in value and is currently worth $100,000. The company announces...
- E Engineering Heellppp pleasssee Explain why the engineer in the following scenario should not have chosen to build a prototype, and recommend the action he should have taken. Ian...
- B Business Assume a company’s equipment carries a book value of $48,000 ($49,500 cost less $1,500 accumulated depreciation) and a fair value of $44,250, and that the $3,750 decline...

Ответ:
(a) 6.57 g of methanol (CH3OH) in 1.50 × 102 mL of solution. It is known that ... 32.042 g/mol. In order to calculate the molarity, 1st we have to find out the number of moles, ... 34P: Balance the following equations and write the corresponding ionic a. ... 38P: (a) Without referring to Figure 4.11. give the oxidation numbers of.
Explanation:
Ответ:
Thus empirical and molecular formula is sa,e for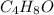
Explanation:
Molecular formula is the chemical formula which depicts the actual number of atoms of each element present in the compound.
Empirical formula is the simplest chemical formula which depicts the whole number of atoms of each element present in the compound.
1. The empirical formula is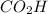
The empirical weight of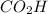 = 1(12)+2(16)+1(1)= 45g.
= 1(12)+2(16)+1(1)= 45g.
The molecular weight = 90 g/mole
Now we have to calculate the molecular formula:
The molecular formula will be=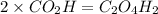
2. The empirical formula is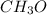
The empirical weight of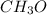 = 1(12)+3(1)+1(16)= 31 g.
= 1(12)+3(1)+1(16)= 31 g.
The molecular weight = 62 g/mole
Now we have to calculate the molecular formula:
The molecular formula will be=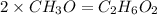
3. The empirical formula is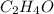
The empirical weight of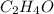 = 2(12)+4(1)+1(16)= 44 g.
= 2(12)+4(1)+1(16)= 44 g.
The molecular weight = 88 g/mole
Now we have to calculate the molecular formula:
The molecular formula will be=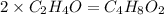
4. The empirical formula is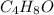
The empirical weight of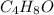 = 4(12)+8(1)+1(16)= 72 g.
= 4(12)+8(1)+1(16)= 72 g.
The molecular weight = 72 g/mole
Now we have to calculate the molecular formula:
The molecular formula will be=