sherlyndaniel89
29.11.2020 •
Chemistry
How many molecules/atoms of excess reactant will remain after the reaction is complete?
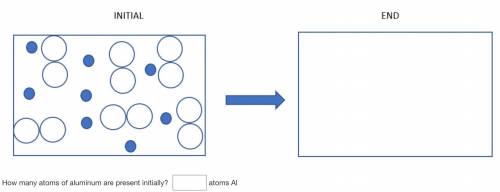
Solved
Show answers
More tips
- C Computers and Internet Boost your processor performance with these easy tips...
- L Leisure and Entertainment When will Maslenitsa start?...
- F Food and Cooking Discovering the Mysterious Fruit of Feijoa...
- B Business and Finance How to Open an Online Store? A Detailed Guide for Beginners...
- W Work and Career How to Write a Resume That Catches the Employer s Attention?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
Answers on questions: Chemistry
- C Chemistry What happens to the radius of an atom when an element loses an electron? what charge desthe element have?...
- C Chemistry Which region of earth s oust experiences the least amount of pressure mante mes core sam...
- C Chemistry 1. Circle the one in each pair represents a greater number of items a) 5 dozen mole of lithium atoms or 60 mole of sulfur atoms b) 0.75 mole of cars or 4.5x1023 cars c) 7701...
- C Chemistry Available Apr 14 at 12am - May 10 at 11:59pm 27 days Using 18.92 g of PF3 and 18.84 g of XeF4 in the following reaction, determine the Limiting Reactant. PF3 + XeF4 --- PF5 +...
- C Chemistry PLEASE HELP MEEE I WILL MARK BRAINLIST FOR THIS ITS SUPER SIMPLE.... all I need you to do its to type out these pictures word for word it pretty simple...
- C Chemistry How do you determine how many boiling stones to use when boiling a solution?...
- C Chemistry Given that there are 1.60 mol of CS2 and 5.60 mol of O2 , which reactant will run out first...
- C Chemistry Veronal, a barbiturate drug, is generally administered as its sodium salt. what is the ph of a solution of nac8h11n2o3 that contains 10. mg of the drug in 250 ml of solution?...
- C Chemistry Astronomers use radio waves to study__...
- C Chemistry Someone please help me im give you so much points ☺️...

Ответ:
Once you have identified the limiting reactant, you calculate how much of the other reactant it must have reacted with and subtract from the original amount.
Explanation:
1. Write the chemical equation.
2.Calculate the moles of product from the first reactant.
3.Calculate the moles of product from the second reactant.
4.Identify the limiting reactant and the excess reactant.
5.Calculate the mass of excess reactant used up.
6.Calculate the mass of unused excess reactant.
7.Calculate the mass of limiting reactant needed to react with the unused excess reactant.
Ответ:
Wind speed largely determines the amount of electricity generated by a turbine. Higher wind speeds generate more power because stronger winds allow the blades to rotate faster. Faster rotation translates to more mechanical power and more electrical power from the generator.
Explanation: