How many molecules of CO2 are contained in 4.40g of CO2
Solved
Show answers
More tips
- F Family and Home How to Remove Tar Stains: Tips and Recommendations from Experts...
- H Health and Medicine Novomin: What is it and how to use it?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
Answers on questions: Chemistry
- M Mathematics Which equation is perpendicular to y= 3/2x + 2 y= -2/3x + 5 y= 2/3x + 7 y= -3/2x + 4 y= 3/2x + 3...
- E English Which of the following is an example of an informal definition? Geneticist J.B.S. Haldane makes an interesting point: I am quite sure that our views on evolution would...
- M Mathematics What does, “What scale and intervals would be appropriate to make a graph of the data” mean...
- P Physics As you are waiting at the Willard airport for a friend coming from Chicago, suddenly something goes wrong with the loudspeakers, and they start making a low pitch...
- B Biology Determine the correct order of the genes on the chromosome, given the following information: Genes A and T cross over at a rate of 18% Genes M and T cross over at...

Ответ:
6.02 x 10²²molecules
Explanation:
Given parameters:
Mass of CO₂ = 4.4g
Unknown:
Number of molecules in CO₂ = ?
Solution:
To find the number of molecules in the given mass, we have to find the number of moles in the compound first;
Number of moles =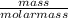
Molar mass of CO₂ = 12 + 2(16) = 44g/mol
Insert the parameters and solve;
Number of moles = = 0.1mol
= 0.1mol
1 mole of a substance contains 6.02 x 10²³molecules
0.1 mole of CO₂ will contain 0.1 x 6.02 x 10²³molecules
= 6.02 x 10²²molecules
Ответ:
7877777
Step-by-step explanation: