4801651052
20.04.2020 •
Chemistry
How many moles are present in 4.7 liters of Argon gas at STP?
Solved
Show answers
More tips
- O Other Discovering the Funniest Joke Ever Told...
- F Food and Cooking How to Make Napoleon Cake: A Step-by-Step Guide...
- F Food and Cooking From Latte to Espresso: Which Coffee Drink is the Most Popular on Earth?...
- C Computers and Internet How to Set Up Internet on iPhone? Detailed Guide with Step-by-Step Instructions...
- F Family and Home How to Teach Your Child to Speak: Tips and Recommendations...
- P Philosophy Agnosticism: Opinion or Belief?...
- S Style and Beauty How to choose the best mascara for your eyelashes...
- F Food and Cooking Discover Delicious Recipes You Can Make with Ground Meat...
- C Computers and Internet Google Search Tips and Tricks: Everything You Need to Know...
- S Science and Technology Why is there no gravity on other planets?...
Answers on questions: Chemistry
- C Chemistry A student has a solution of 2.0 M MgSO4. What volume (in mL) of the original solution is needed to make 100 mL of 0.40 M MgSO4? A) 20 B) 50 C) 200 D) 500...
- C Chemistry Mars has a mass of 642,000,000,000,000,000,000,000 kg. How would scientists express this number in scientific notation?...
- C Chemistry In 1906, a 600 g diamond was found at the Premier Mine in South Africa- the world s largest uncut diamond. Suppose the diamond s temperature changed by 2℃ as it was cut. If...
- C Chemistry How much salt is in 300 g of a 3.5 % solution of salt water?...
- C Chemistry A student has the following data: V1 = 822 mL; T1 = 75.0 °C; T2 = 25.0 °C. She uses this data to calculate V1 and gets -274 mL. Is her calculation correct? Choose the best...
- C Chemistry Calculate ΔS°rxn (J/k) for 3NO2(g) + H2O(l)LaTeX: \longrightarrow⟶ NO(g) +2HNO3(l) C6H12O6(s) + 6O2(g) LaTeX: \longrightarrow⟶ 6H2O(g) +6CO2(g) Enter numbers to 1 decimal places....
- M Mathematics A sphere is inscribed in a cube with a volume of 27 cubic inches. What is the volume of the sphere? Round your answer to the nearest whole number. The volume of the sphere...
- E English The second paragraph of the Declaration of Independence establishes general principles about the rights of man and the role of government. What is the most likely reason that...
- H History How did railroads impact Texas?...
- M Mathematics Click an item in the list or group of pictures at the bottom of the problem and, holding the button down, drag it into the correct position in the answer box. Release your...

Ответ:
The answer for the following problems is mentioned below.
Therefore the number of moles present in Argon is 0.20 moles.Explanation:
Mole:
The mass of a substance containing the same number of fundamental units as there are atoms in exactly 12.000 g of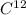 .
.
Given:
volume (v) = 4.7 litres
Volume of argon at STP conditions = 22.4 litres
To find:
volume of argon at STP conditions
We know;
n =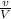
where;
n represents the no of moles
v represents the volume of the argon
V represents the volume of argon at STP
volume of the argon at STP conditions is 22.4 litres
So;
n =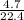
n = 0.20 moles
Therefore the number of moles present in Argon is 0.20 moles.
Ответ:
Electrons will be gained