powella033
03.11.2020 •
Chemistry
How many moles are present in
14g of Nitrogen atom and 14g of Nitrogen molecules
Solved
Show answers
More tips
- C Computers and Internet Отправляем смс через интернет: легко и просто...
- L Leisure and Entertainment The Best Film of 2010: A Look Back at the Academy Awards...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- L Leisure and Entertainment What to Bring on a Hike? Essential Items to Pack for a Safe and Enjoyable Adventure...
- L Leisure and Entertainment Couchsurfing: A New Way to Travel...
- S Style and Beauty Autotanning: Harmful or Safe?...
- F Food and Cooking 10 Ideas for a Wedding Anniversary Gift...
- H Health and Medicine How to Reduce Sweating in the Heat and Beyond: Say Goodbye to Excessive Sweat...
- F Food and Cooking Do Aphrodisiacs Really Work? Separating Fact from Fiction...
- H Health and Medicine What to Eat to Lose Weight?...
Answers on questions: Chemistry
- C Chemistry A constant volume of oxygen is heated from 125°C to 175°C. The initial pressure is 4.1 atm. What is the final pressure?...
- C Chemistry A population of white moths moves into an area dominated by grey moths. How do you think the introduction of white moths will impact the grey moths?Explain your answer....
- C Chemistry According to the reaction below, how many grams of KCl (potassium chloride) can be formed from 70.90 g of chlorine with excess potassium? 2K + Cl2 --- 2 KCl...
- C Chemistry An atom that has one or two valence electrons is most likely to be - Nonreactive only A) with nonmetals B) Reactive with metals C) Highly reactive D) nonreactive...
- C Chemistry What is an example of a place formed by hot-spot valcanoes? a.caymen islands b. florida keys c. australia d. hawaiin islands...
- C Chemistry The ir spectrum of which type of compound generally exhibits evidence of hydrogen bonding?....
- C Chemistry Consider the following four solutions: a) 0.10 M HA b) 0.10 M KA (potassium salt of A1-) c) 0.10 M HB d) 0.10 M KB Assume that pKa of HA is greater than that of HB. The solution...
- C Chemistry In a 0. 1 m solution of glycine at ph 9. 0, what fraction of glycine has its amino group in the form?....
- C Chemistry What is the smallest alkene that has enantiomers but no cis/trans isomers? show them with their respective names....
- C Chemistry An electron moves along the z-axis with vz=6. 0×107m/s. As it passes the origin, what are the strength and direction of the magnetic field at the following (x, y, z) positions?....

Ответ:
The mass of carbon and hydrogen in the sample is 0.1087 g and 0.0066 g respectively and the percentage composition of carbon and hydrogen in the sample is 94.27 % and 5.72 % respectively.
Explanation:
The chemical equation for the combustion of hydrocarbon having carbon and hydrogen follows:
where, 'x' and 'y' are the subscripts of carbon and hydrogen respectively.
We are given:
Mass of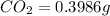
Mass of
Mass of sample = 0.1153 g
We know that:
Molar mass of carbon dioxide = 44 g/mol
Molar mass of water = 18 g/mol
For calculating the mass of carbon:In 44g of carbon dioxide, 12 g of carbon is contained.
So, in 0.3986 g of carbon dioxide,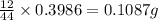 of carbon will be contained.
of carbon will be contained.
For calculating the mass of hydrogen:In 18g of water, 2 g of hydrogen is contained.
So, in 0.0578 g of water,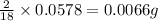 of hydrogen will be contained.
of hydrogen will be contained.
To calculate the percentage composition of a substance in sample, we use the equation:
 ......(1)
......(1)
For Carbon:Mass of sample = 0.1153 g
Mass of carbon = 0.1087 g
Putting values in equation 1, we get:

For Hydrogen:Mass of sample = 0.1153 g
Mass of hydrogen = 0.0066 g
Putting values in equation 1, we get:
Hence, the mass of carbon and hydrogen in the sample is 0.1087 g and 0.0066 g respectively and the percentage composition of carbon and hydrogen in the sample is 94.27 % and 5.72 % respectively.