How many moles of Csl must be dissolved into 800 ml of a 0.40 M
solution?
Solved
Show answers
More tips
- S Style and Beauty How to Get Rid of a Double Chin?...
- F Food and Cooking How to Cook Julienne? Recipes and Tips...
- D Dating, Love, Relationships 10 Useful Tips on How to Survive a Breakup?...
- F Food and Cooking Apple Cider Vinegar: The Ultimate Health and Beauty Solution...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
Answers on questions: Chemistry
- M Mathematics Selected QUESTION 17 Of 20If 15 men dig a trench in 10 days, how many men will be required to dig the trench for 2 days?...
- G Geography The warmer waters usually mean bad news for fisherman and for birds—fewer cold-water fish in the region. . [according to conrad lautenbacher of the national oceanic and atmospheric...
- A Arts What is a meander pattern? which two civilizations mainly used this technique?...

Ответ:
0.32 moles of Csl must be dissolved into 800 ml of a 0.40 M solution.
Explanation:
Data given:
volume of the Csl solution = 800 ml or 0.8 litres
molarity of the solution = 0.40 M
number of moles of Csl = ?
molarity is calculated by using the formula:
molarity =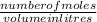
rearranging the equation to get the number of moles:
number of moles = molarity x volume in litres
putting the values in the equation:
number of moles = 0.4 x 0.8
number of moles of Csl in the solution is 0.32
therefore 0.32 moles of Csl should be dissolved in 800 ml of the solution to obtain molarity of 0.4 M.
Ответ:
It is both accurate and precise.
Explanation:
Precision and accuracy are two different terms used to describe data or measurements. Accuracy refers to how close a set of measurements/experimental values is to an accepted or correct value while Precision refers to how close a series of experimental values are to one another.
In the given set of data in the question below, the Correct Value is 59.2 while the experimental values are as follows;
Trial 1: 58.7
Trial 2: 59.3
Trial 3: 60.0
Trial 4: 58.9
Trial 5: 59.2
Based on comparison, it can be observed that these experimental values are close to the correct value (59.2). Hence, they are said to be ACCURATE. Also, the experimental values are close to one another, hence, they are said to be PRECISE.
Therefore, the data set is both accurate and precise.