WhiteWinterRose
14.07.2019 •
Chemistry
How many moles of magnesium hydroxide are needed to produce 74.7 milliliters of water, if the density of water is 0.987 g/ml? show all steps of your calculations as well as the final answer. hno3 + mg(oh)2 → h2o + mg(no3)2?
Solved
Show answers
More tips
- A Auto and Moto How to Deregister a Car in Moscow?...
- F Food and Cooking How to Cook Julienne? Recipes and Tips...
- D Dating, Love, Relationships 10 Useful Tips on How to Survive a Breakup?...
- F Food and Cooking Apple Cider Vinegar: The Ultimate Health and Beauty Solution...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
Answers on questions: Chemistry
- C Chemistry Question 19 unsaved which of the following is not one of the warning signs used to predict a volcanic eruption? 1) changing chemistry of volcanic gas 2) development and widening...
- C Chemistry In which item are fuel cells more commonly used? laptops flashlights solar panels propane lanterns...
- C Chemistry Question 14 unsaved which of the following statements is true? 1) as temperature decreases and salinity increases, density increases. 2) as temperature increases and salinity decreases,...
- C Chemistry Energy cannot be created or destroyed. therefore, in order to provide electric energy for a portable laptop, voltaic cells need to be stored in hot places so that they can absorb...
- C Chemistry How many moles of UF6 would have to be decomposed to provide 1.25 moles of fluorine?...
- C Chemistry The fossils found in the bottom layers of sedimentary rock are probably?...
- C Chemistry (b) Which of the following is a mixture? A.Sodium chloride solution B.Sodium chloride C.Sodium D.Chlorine...
- C Chemistry What volume would 9.5 L of nitrogen dioxide (NO2) gas at a pressure of 750 torr and a temperature of 22.0 OC occupy at STP? 8.7L 323 mL 323 L 8.7 ml...
- C Chemistry 16. nuclear decay is a random event and a. is independent of other energy influences c. can be sped up upon cooling b. can be sped up upon heating d. can be sped up at room temperature...
- C Chemistry Which source is most likely an authority on the subject? Dr. Vale, a pediatrician, discusses electrical circuitry on her home-improvement website. Jonah, an amateur cook, teaches...

Ответ:
Given volume of water produced in the reaction = 74.7 mL
Density of water = 0.987 g/mL
Calculating the mass of water =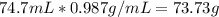
Moles of water produced =
The balanced chemical equation between nitric acid and magnesium hydroxide is,
1 mol Magnesium hydroxide gives 2 mol water.
Moles of Magnesium hydroxide =
Ответ: