Pomskis2341
08.05.2020 •
Chemistry
How many moles of magnesium nitrate are in 6.35 grams of magnesium nitrate?
Solved
Show answers
More tips
- S Sport How to Do Push-ups Correctly?...
- S Style and Beauty How to Grow Hair Faster: Real Methods and Advice...
- F Family and Home How to Remove Fading from Clothes: Tips and Tricks...
- F Food and Cooking How to Make Polendwitsa at Home?...
- F Family and Home Parents or Environment: Who Has the Most Influence on a Child s Upbringing?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
Answers on questions: Chemistry
- M Mathematics Solve each equation or formula for the variable indicated ab+3c=2x, for b...
- H History Governor during whose administration the Texas state legislature expanded juries and grand juries to include women...
- B Biology Explain briefly the forces that govern the upward movement of water and dissolved mineral salts in plants. tom of classification....

Ответ:
0.0428 moles
Explanation:
6.35 g Mg(NO3)2 ×
6.35 g / 148.32 g
0.0428 moles
Ответ:
The tempearture of 11.2 L of CO is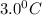 .
.
Explanation:
To calculate the final volume of the system, we use the equation given by Charles' Law. This law states that volume of the gas is directly proportional to the temperature of the gas at constant pressure.
Mathematically,
where,
are the final volume and temperature of the gas.
We are given:
Putting values in above equation, we get:
Thus the tempearture of 11.2 L of CO is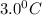 , pressure remaining constant.
, pressure remaining constant.