How many moles of Mg(OH)2 will react with 2.5 mol HCI?
Solved
Show answers
More tips
- C Computers and Internet Boost your processor performance with these easy tips...
- L Leisure and Entertainment When will Maslenitsa start?...
- F Food and Cooking Discovering the Mysterious Fruit of Feijoa...
- B Business and Finance How to Open an Online Store? A Detailed Guide for Beginners...
- W Work and Career How to Write a Resume That Catches the Employer s Attention?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
Answers on questions: Chemistry
- B Biology Hot coffee in a mug over time and the mug warms up. which describes the energy in this system...
- M Mathematics Chandrika made four barrettes. the lengths of the barrettes are 2.8 cm, 3.1 cm, 2.5 cm, and 3 cm.what is the length of the shortest barrette she made? 2.8 cm3.1 cm2.5 cm3...
- B Biology Describe the heat transfer cycle between the sun, the earth, and the earth’s atmosphere....

Ответ:
1.25mole of Mg(OH)₂
Explanation:
The reaction is between Mg(OH)₂ and HCl, this is a neutralization reaction between an acid and a base.
Mg(OH)₂ + 2HCl → MgCl₂ + 2H₂O
The balanced reaction equation is given above.
2 mole of HCl reacts with 1 mole of Mg(OH)₂
So, 2.5mole of HCl will react with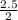 = 1.25mole of Mg(OH)₂
= 1.25mole of Mg(OH)₂
The number of moles of Mg(OH)₂ is given as 1.25mol
Ответ: