How many moles of n are in 0.221 g of n2o?
Solved
Show answers
More tips
- S Sport Where can you play football in Moscow?...
- F Food and Cooking 10 Reasons Why You Should Avoid Giving Re-Gifts: An Informative Guide...
- S Sport How to wrap boxing hand wraps? Everything you need to know!...
- A Animals and plants 5 Tips for Taking Care of Yews to Keep Them Green and Beautiful...
- H Health and Medicine How to Calm Your Nerves? Expert Tips That Actually Work...
- O Other What is a Disk Emulsifier and How Does it Work?...
- S Sport How to Pump Your Chest Muscle? Secrets of Training...
- C Computers and Internet How to Get Rid of 3pic Infector: Everything You Need to Know...
- S Style and Beauty How to Grow Hair Faster: Real Methods and Advice...
- C Computers and Internet How to Top Up Your Skype Account Without Losing Money?...
Answers on questions: Chemistry
- C Chemistry Problem page gaseous butane will react with gaseous oxygen to produce gaseous carbon dioxide and gaseous water . suppose 5.2 g of butane is mixed with 32.6 g of oxygen. calculate...
- C Chemistry 17 What is the volume of 2.00 moles of nitrogen gas (N2) at 1 atm and 0 °C? (R=0.0821, when using atm for pressure)...
- C Chemistry In the reaction CaCO3+2HCl CaCl 2 +H 2 O+CO 2 how many liters of CO 2 can be produced from 45.0g of calcium carbonate?...
- C Chemistry The overall electrical charge on a stable element is...
- C Chemistry How many total number of atoms are in the compound KMO4?...
- C Chemistry Which body system brings in oxygen from the environment, and how do body cells use the oxygen to release energy?...
- C Chemistry This function option is not found under the function icon located on the toolbar...
- C Chemistry Calculate the angle of impact for a blood drop with a length of 16.4 mm and a width of 10.1 mm. 53° 38° 62° 1.62...
- C Chemistry How can we make compost by piling organic materials?it s argent plz someone help me...
- C Chemistry How are molecules different from that of gas explain how they look at the molecular level...

Ответ:
0.221 g of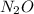 have 0.010 mol of N
have 0.010 mol of N
Explanation:
Mass of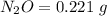
Step 1
Calculation of moles of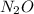
Molecular mass of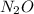 = 14 × 2 + 16 = 44
= 14 × 2 + 16 = 44
Calculation of moles of N
1 mol of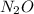 has 2 moles of N
has 2 moles of N
0.005 mol of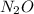 will have
will have
= 0.005 × 2 = 0.010 mol of N
So, 0.221 g of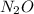 have 0.010 mol of N
have 0.010 mol of N
Ответ:
hypoglycemia
Explanation:
In general, hypoglycemia is a blood sugar level less than 70 mg/dL. If you have diabetes, your care team will give you specific blood sugar level goals. So, your low blood sugar number may be different from someone without diabetes.. The most common cause of hypoglycemia is a side effect of diabetes treatment.