How much energy, in joules, is needed to heat 100.0 g of ethyl alcohol from 22.0°C to 60°C? The specific heat of ethyl alcohol is 2.44 J/g°C.
Solved
Show answers
More tips
- F Food and Cooking Homemade kvass: recipe and brewing process...
- O Other What is a Disk Emulsifier and How Does it Work?...
- H Health and Medicine How to Calm Your Nerves? Expert Tips That Actually Work...
- A Animals and plants 5 Tips for Taking Care of Yews to Keep Them Green and Beautiful...
- S Sport How to wrap boxing hand wraps? Everything you need to know!...
- F Food and Cooking 10 Reasons Why You Should Avoid Giving Re-Gifts: An Informative Guide...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Style and Beauty How Are Eyelash Extensions Applied? All Your Questions Answered...
- F Food and Cooking 10 Tips for Proper Sushi Consumption...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
Answers on questions: Chemistry
- C Chemistry Is wrought iron substitutional or interstitial?...
- C Chemistry What and where is silver nitrate used...
- C Chemistry 15.45 grams of solid potassium chlorate decomposes when heated to form solid potassium chloride and oxygen gas. Assuming 100% yield, how many grams of solid potassium chloride...
- C Chemistry A buffer is made by mixing 60.0 mL of 0.5 M acetic acid and 40.0 mL of 0.50 M sodium acetate. What is the pH of the buffer solution?...
- C Chemistry Using s, p, d, f notation, what is the electron configuration for a neutral atom of magnesium?...
- C Chemistry How many grams of sodium oxalate do you need to drink to make 20 g kidney stone (calcium oxalate)?...
- C Chemistry Which statement explains why the element carbon forms so many compounds? 1 Carbon atoms combine readily with oxygen 2 Carbon atoms readily attract electrons. 3 Carbon readily...
- C Chemistry Balance the following equation with the smallest whole number coefficients. Which of the following is the sum of the coefficients in the balanced equation? PtCl4 + XeF2➡️PF6...
- C Chemistry A student analyzes the composition of steel and finds that some of the elements in different samples of steel are found to be in different proportions. Classify the type...
- C Chemistry What happens when balloon filled with CO2 is released in air...

Ответ:
The energy is 9,272 J (Joules)
Explanation:
Calorimetry is the measurement and calculation of the amounts of heat exchanged by a body or a system.
In this way, there is a direct proportional relationship between heat and temperature (Two magnitudes are directly proportional when there is a constant so that when one of the magnitudes increases, the other also; and the same occurs when one of the two decreases .). The constant of proportionality depends on the substance that constitutes the body as on its mass, and is the product of the specific heat for the mass of the body. So, the equation that allows calculating heat exchanges is:
Q = c * m * ΔT
Where Q is the heat exchanged by a body of mass m, made up of a specific heat substance c and where ΔT is the temperature variation
In this case:
Q= ?c= 2.44Replacing:
Q= 2.44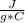 *100 g* 38 °C
*100 g* 38 °C
Solving:
Q=9,272 J
The energy is 9,272 J (Joules)
Ответ:
2.) Notes of the chart
3.) chart symbols
4.) chart corrections
5.) GPS positions
6.) radar fixes
7.) Visual fixtures and position circle and position line