heyItsLuna234
31.07.2019 •
Chemistry
How much energy is evolved during the formation of 197 g of fe, according to the reaction below? fe2o3(s) + al(s) → al2o3(s) + fe(s) δh°rxn = -852 kj?
Solved
Show answers
More tips
- W Work and Career How to Start Your Own Business: Tips and Recommendations...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...

Ответ:
Chemical reaction: Fe₂O₃(s) + Al(s) → Al₂O₃(s) + Fe(s); ΔH°rxn = -852 kJ.
m(Fe) = 197 g.
n(Fe) = m(Fe) ÷ M(Fe).
n(Fe) = 197 g ÷ 55,85 g/mol.
n(Fe) = 3,527 mol.
Use proportion: 2 mol(Fe) : -854 kJ = 3,527 mol(Fe) : ΔH°rxn.
ΔH°rxn = -854 kJ · 3,527 mol ÷ 2 mol.
ΔH°rxn = -1510 kJ = -1,51·10³ kJ.
Ответ:
Answer : The energy evolved in the reaction is 1499.52 kJ
Explanation : Given,
Mass of Fe = 197 g
Molar mass of Fe = 56 g/mole
Enthalpy of reaction = -852 kJ
First we have to calculate the moles of Fe.
Now we have to calculate the energy evolved in the reaction.
From the balanced chemical reaction we conclude that,
As, 2 mole of Fe evolved energy = 852 kJ
So, 3.52 mole of Fe evolved energy =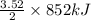
= 1499.52 kJ
Therefore, the energy evolved in the reaction is 1499.52 kJ
Ответ: