hhgjhbmbhg266
03.09.2019 •
Chemistry
How much heat is produced when 100ml of 0.25 m hcl (density 1.00g/ml) and 200 ml of 0.150 m naoh (densty 1.00g/ml) are mixed?
hcl + naoh ? nacl + h2o ho298= -58kj
if both solutions are the same temperatureand heat capaciy of the products is 4.19 j/gc, how much will the temperature increase? what assumption did you make in your calculation?
Solved
Show answers
More tips
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- L Leisure and Entertainment Carving: History and Techniques for Creating Vegetable and Fruit Decorations...
- P Photography and Videography How to Choose the Perfect Photo Paper for Your Images?...
- H Health and Medicine What vaccines do children need?...
- H Health and Medicine Reasons for the Appearance of Warts: Everything You Need to Know...
- A Art and Culture How to Learn Screaming: Step-by-Step Guide for Beginners...
Answers on questions: Chemistry
- H History The followers of chiang kaishek in China were known as A; communist B: boxers C;nationalists D; fascists...
- E English For each of the following lines from “Brown Girl Dreaming,” identify the nouns. Then, label each noun as common, proper, or possessive. Note: There may be more than...
- A Arts Hey guys dis meh new oc -w-...
- M Mathematics What must the length of segment ad be for the quadrilateral to be a parallelogram? 8 units 16 units 31 units 62 units...

Ответ:
Explanation:
The given data is as follows.
For HCl : Volume = 100 mL, Density = 1.00 g/ml, Molarity = 0.25 M
For NaOH : Volume = 200 mL, Density = 1.00 g/ml, Molarity = 0.150 M
Therefore, mass of HCl will be calculated as follows.
Density =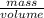
1.00 g/ml =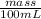
mass = 100 g
Also, number of moles =
Hence, no. of moles of HCl = (as 1 ml = 1000 L)
(as 1 ml = 1000 L)
=
On the other hand, mass of NaOH will be calculated as follows.
Density =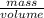
1.00 g/ml =
mass = 200 g
Also, number of moles =
Hence, no. of moles of HCl = (as 1 ml = 1000 L)
(as 1 ml = 1000 L)
=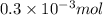
Total mass = (100 g + 200 g) = 300 g
Since, HCl is the limiting reagent over here. So, heat produced by will be calculated as follows.
will be calculated as follows.
Q =
=
= kJ
kJ
=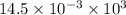 J
J
Also, it is known that relation between Q and temperature change is:
Q =
Hence, putting the values into the above formula as follows.
Q =
14.5 J =
Thus, we can conclude that increase in temperature is .
.
Ответ: