Hydrogen iodide is not produced by the same method is for hydrogen chloride why with reaction
Solved
Show answers
More tips
- P Philosophy How did the concept of module arise in computer science?...
- D Dating, Love, Relationships Why Should the Man be Active and the Woman Passive during Foreplay?...
- S Society and Politics Выборы: Смысл, Значение и Отражение...
- B Business and Finance How to Get Your Money Back When Lending? Top 7 Ways...
- F Family and Home Do Lullabies Help Babies Sleep or Is it Just a Myth?...
- F Family and Home Why Does God Punish Us Constantly and How Can We Fix It?...
- D Dating, Love, Relationships Why do we feel shame?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- O Other How to Accidentally Get a Rare Coin with Your Change and How to Know Its Value?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
Answers on questions: Chemistry
- E English Watching funny videos join the meeting the code and on other question...
- M Mathematics Convert 40 pounds to kilograms. (the conversion factor is 2.2 pounds/kilogram. round to the nearest hundredth.) a. 0.05 kilograms b. none of these c. 18.18 pounds d. 18.18 kilograms...
- P Physics When an antigen attacks the body, it activates adaptive immunity. this adaptive immunity produces a strong response when it encounters the same antigen for the second time. which feature...
- M Mathematics Kelby has several pets. She has B birds, C cats, and F frogs. The expression B + (C+F) represents the total number of pets Kelby has. What does C + F represent in this context? Choose...
- M Mathematics Consider the fourier sine series of each of the following functions. in this exercise do not compute the coefficients but use the general convergence theorems (theorems 2, 3, and 4)...

Ответ:
Using Phosphoric acid will work perfectly for producing Hydrogen halides because its not an Oxidizing agent. ...
Using an ionic chloride and Phosphoric acid
H3PO4 + NaCl ==> HCl + NaH2PO4
H3PO4 + NaI ==> HI + NaH2PO4
H2SO4 + NaCl ==> HCl + NaHSO4
This method(Using H2So4) will work for all hydrogen hydrogen halide except Hydrogen Iodide and Hydrogen Bromide.
The Sulphuric acid won't be useful for producing Hydrogen Iodide because its an OXIDIZING AGENT. Whist producing the Hydrogen Iodide... Some of the Iodide ions are oxidized to Iodine.
2I-² === I2 + 2e-
Ответ:
Answer : The correct option is, (A)
Explanation :
Empirical formula : It is the simplest form of the chemical formula which depicts the whole number of atoms of each element present in the compound.
Molecular formula : it is the chemical formula which depicts the actual number of atoms of each element present in the compound.
For determining the molecular formula, we need to determine the valency which is multiplied by each element to get the molecular formula.
The equation used to calculate the valency is :
As we are given that the empirical formula of a compound is and the molar mass of compound is, 180.2 gram/mol.
and the molar mass of compound is, 180.2 gram/mol.
The empirical mass of = 1(12) + 2(1) + 1(16) = 30 g/eq
= 1(12) + 2(1) + 1(16) = 30 g/eq
Molecular formula =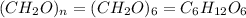
Thus, the molecular formula of the compound will be,