tacojordan4128
18.01.2021 •
Chemistry
I'd love some help please thank you

Solved
Show answers
More tips
- H Health and Medicine Is it true that working with a computer is harmful to your eyesight?...
- S Sport When is the Champions League final?...
- H Health and Medicine Is Folic Acid a Necessary Vitamin?...
- W Work and Career How to Start Your Own Business: Tips and Recommendations...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
Answers on questions: Chemistry
- C Chemistry Which of these statements is false? A. Atoms of different elements can combine to form compounds. B. Oxygen and copper are examples of elements. C. Atoms of different elements...
- M Mathematics Giving 50 points for both pages + brainlest for first answer...
- M Mathematics Q = 3x + 9x? idk this one lol...
- P Physics I NEED HELP PLEASE ANSWER CORRECTLY. I LL WILL GIVE YOU A BRAINLIEST ANSWER IF YOU RE ANSWER IS CORRECT........ PROMISE :) QUESTION:Q1. Where was the location of3 tropical storm...
- M Mathematics Cipher code LT YT UFWNX Please decode it for me...
- H History What are the best answers, check all that apply...

Ответ:
Because that answer makes the most sense.
Ответ:
Explanation:
We have to start with the reaction:
We have the same amount of atoms on both sides, so, we can continue. The next step is to find the number of moles that we have in the 110.0 g of carbon dioxide, to this, we have to know the atomic mass of each atom:
C: 12 g/mol
O: 16 g/mol
Mg: 23.3 g/mol
If we take into account the number of atoms in the formula, we can calculate the molar mass of carbon dioxide:
In other words: . With this in mind, we can calculate the moles:
. With this in mind, we can calculate the moles:
Now, the molar ratio between carbon dioxide and magnesium carbonate is 1:1, so:
With the molar mass of (
(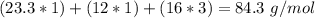 . With this in mind, we can calculate the grams of magnesium carbonate:
. With this in mind, we can calculate the grams of magnesium carbonate:
I hope it helps!