I need help on this, It’s due today. Please, anybody
Solved
Show answers
More tips
- F Family and Home How to Remove Fading from Clothes: Tips and Tricks...
- F Family and Home How to Remove Tar Stains: Tips and Recommendations from Experts...
- L Leisure and Entertainment How to Learn to Draw Graffiti: Tips for Beginners...
- G Goods and services How to sew a ribbon: Tips for beginners...
- L Leisure and Entertainment How to Land on the Moon: Your Comprehensive Guide...
- H Health and Medicine Novomin: What is it and how to use it?...
- A Art and Culture Attention, the Final Episode of Margo is Almost Here!...
- A Art and Culture How to Learn Screaming: Step-by-Step Guide for Beginners...
- D Dating, Love, Relationships 10 Useful Tips on How to Survive a Breakup?...
- S Style and Beauty How to Break in New Shoes: 7 Simple Methods...
Answers on questions: Chemistry
- B Business The following information is available for a company s utility cost for operating its machines over the last four months. Month Machine hours Utility cost January 1,020 $6,570...
- M Mathematics Cathy has to deposit $410 worth of five- and tendollar bills. she has 1 fewer than three times as many tens as she does five-dollar bills. how many of each bill does she have to...
- E English You re friends with those people ? those people are a)negative b)neutral c)postivie...
- S Social Studies what initiative are you implementing to ensure that children uses social media platforms appropriately...

Ответ:
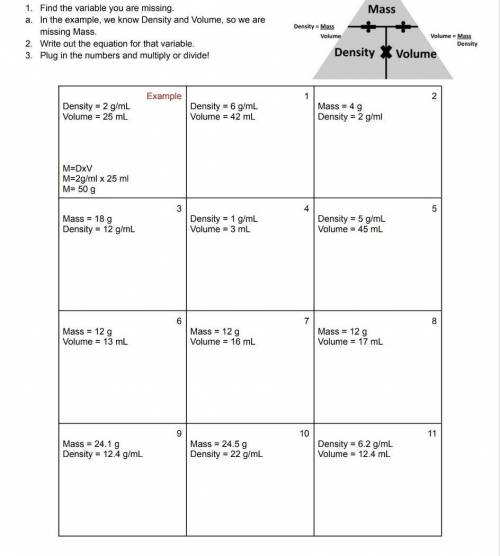
1 = 252g, 2 = 2mL, 3 = 1.5mL, 4 = 3g, 5 = 225g, 6 = 0.92g/mL, 7 = 0.75g/mL, 8 = 0.71g/mL, 9 = 1.9mL, 10= 1.11mL, 11 = 76.9g
Explanation:
This problem is testing how well you can move around the equation D = m/v where D = Density (g/mL), m= mass of sample (g), v = volume of sample (mL).
Ответ:
The ratio of mass of sulfur atom to mass of oxygen atom in sulfur dioxide is 1: 1.
Explanation:
Law of constant proportion states that In a chemical substance the elements are always present in definite proportions by mass. This law is also known as 'Law of definite proportions '.
Mass of 1 atom of sulphur = 32 g
Mass of 1 atom of oxygen = 16 g
Mass of 2 atoms of oxygen =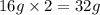
In formation of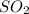 , 1 atom of sulfur combines with 2 atoms of oxygen and thus the mass ratio will be 32: 32= 1:1 .
, 1 atom of sulfur combines with 2 atoms of oxygen and thus the mass ratio will be 32: 32= 1:1 .
Thus the ratio of mass of sulfur atom to mass of oxygen atom in sulfur dioxide is 1: 1.