Identify the element oxidized and the element reduced. Give the number of electrons lost or gained by each atom. Li + H2O → LiOH + H2
Solved
Show answers
More tips
- C Computers and Internet Best Antivirus: How to Choose Protection for Your Computer?...
- L Legal consultation What Documents Are Required for a Russian Passport?...
- S Sport How to Choose Tennis Rackets?...
- H Health and Medicine AKDS Vaccination: Ensure Your Child s Safety...
- H Health and Medicine Naskol ko Opasen Ukus Kleshcha i Kak Ego Raspoznat...
- C Computers and Internet How to Delete Your Account on Odnoklassniki...
- H Health and Medicine What to Do When Your Jaw Locks Up?...
- G Goods and services What Are the Most Popular Services?...
- P Philosophy How did the concept of module arise in computer science?...
Answers on questions: Chemistry
- C Chemistry Florida can be described as a peninsula, surrounded on three sides by salt water: the Atlantic Ocean and the Gulf of Mexico. The low lying areas of Florida are particularly...
- C Chemistry The fictional element “Nt” contains atoms with two valence electrons. Which type of intermolecular force is most likely responsible for the properties of NtF2? O...
- C Chemistry write a chemical equations for the reaction that takes place between each of the following paris of reactants...
- C Chemistry What are 4 technologes science created...
- C Chemistry The pressure above a pure sample of solid Substance X at 7. C is lowered. At what pressure will the sample melt...
- C Chemistry Does nitrogen triiodide dipole?....
- C Chemistry The frequency of a sound wave is 457 Hz and the speed is 342.5 m/s. What is the sound’s wavelength...
- C Chemistry Find ΔS o for the combustion of ammonia to nitrogen dioxide and water vapor. J/K Is this the sign of ΔS o you would get if you only considered the change in moles...
- C Chemistry Which choice represents a pair of resonance structures? note that lone pairs have been omitted for clarity. view available hint(s) which choice represents a pair...
- C Chemistry Alab director asks for two students, lydia and damien, to each select a bottle of a concentrated weak base. when they reach the cabinets, they find different chemical...

Ответ:
A)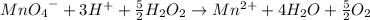
B) 7.5 molar
Explanation:
A) Reduction
Oxidation
Multiplying the oxidation reaction by 5/2 and adding it to the reduction equation:
+
-----------------------------------------------------------------------------------------------------
B) 10 ml = 0.01 L
20 ml = 0.02 L
mol of MnO4− = molarity*volume = 1.5*0.02 = 0.03
1 mol of MnO4− reacts with 5/2 mol of H2O2, then:
mol of H2O2 = 0.03*5/2 = 0.075
molarity = mol/volume = 0.075/0.01 = 7.5 molar