If 32.146g of mgso4 is added to enough water to make 100.00ml solution, what is the molarity of mgsoa? 1) 2.6706 m 2) 3.7445m 3) 0.037445 4) 0.0026706 m 5) 3869.4 m
Solved
Show answers
More tips
- A Animals and plants How to Properly Care for Orchids: Secrets to Successful Cultivation...
- W Work and Career Which types of activities are not subject to mandatory licensing?...
- C Computers and Internet Thin Client: What It Is and Why You Need It?...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- C Computers and Internet Clearing Cache: How to Speed Up Your Browser...
- S Style and Beauty How are artificial nails removed?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- F Food and Cooking Everything You Need to Know About Pasta...
Answers on questions: Chemistry
- C Chemistry The temperature of a sample of ammonia gas is changed without changing its volume, causing a change in pressure from 9.2174 atm to 9.8958 atm. If the starting temperature...
- C Chemistry If the temperature is raised which direction will the equilibrium shift? 1 N,+ 3 H2 2 NH3 + heat A) Right towards the product to remove the added heat B)Left towards...
- C Chemistry A sample of krypton gas occupies a volume of 8.96L at 48.0 celcius and 1.08atm. If it is desired to decrease the volume of the gas sample to 7.71 L while decreasing...
- C Chemistry A compound with a molar mass of about 28 g/mol contains 85.7% carbon and 14.3% hydrogen by mass. Write the Lewis structure for a molecule of the compound....
- C Chemistry What is the approximate vapor pressure when the gas condenses at 70°C? 150 760 380 5...
- C Chemistry What type of rock is this?...
- C Chemistry It has been determined that the body can generate 5500 kj of energy during one hour of strenuous exercise. perspiration is the body’s mechanism for eliminating...
- C Chemistry Breathing air that contains 4.0% by volume co2 over time causes rapid breathing, throbbing headache, and nausea, among other symptoms. you may want to reference...
- H History True or False: All countries are legally bound to survey and monitor for disease within their borders....
- C Chemistry PLEASE HELP...with single replacement reaction w/ activity series WILL MARK BRAINLIEST IF HELPFUL Gold IV iodide + Bromine yields to... Thank You!...

Ответ:
Explanation:
Hello,
At first, the moles of magnesium sulfate must be computed by knowing that its molecular mass has a value of 120.366 g/mol:
Now, by knowing that the volume must be in L, it turns out into 0.10000 L, thus:
Best regards.
Ответ:
The first thing we have to do is change and state all the units so that we can use our ideal gas law equation (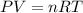 ).
).
650 mmHg is a pressure unit, we have to convert this to kiloPascals. We know that 760 mmHg gives us 101 kPa.
P = 86kPa
T = 15°C + 273K = 288K
R (Gas constant) = 8.31 kj/mol
Molar mass of Ammonia (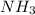 ) = (1 x 3) + (14) = 17g/mol
) = (1 x 3) + (14) = 17g/mol
n (moles) =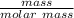
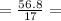 3.34 mol
3.34 mol
V = ?
Rearrange the equation to solve for Volume:
Substitute the values inside:
V =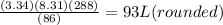
Therefore 93 L of volume is occupied by the ammonia gas.