laurenrubin18
27.06.2019 •
Chemistry
If 58.67g of mercuric oxide were completely decomposed to generate 54.34 g of mercury how many grams of oxygen should have been produced
Solved
Show answers
More tips
- A Auto and Moto Discovering the Leader: What is the Most Expensive Car in the World?...
- H Health and Medicine Hangover: How to Get Rid of It Quickly?...
- S Style and Beauty How to Choose the Right Fur Coat and Avoid Regrets?...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- C Chemistry HELLO, DONT ANSWER THIS ITS FOR MY FRIEND...
- C Chemistry Please help me answer this I will give a lot of points...
- C Chemistry How many grams is 2.11 moles of NaCO3?...
- C Chemistry What is the pH of a 0.00530 M solution of HCI?...
- C Chemistry Someone plz help with this...
- C Chemistry Can somebody please help me!!...
- C Chemistry Draw Lewis dot symbols for the following species: a) Lib) Tic) Znd) Sie) Nef) Mng) Niwh) Agi) Ba) Pb...
- C Chemistry In an aqueous solution at 25°C, if [H30+] = 3.3 * 10^4 M, then [OH-] is:...
- C Chemistry Keystone species are those that are a necessary component to the stability of the ecosystem, and without them, many other organisms would not survive and the ecosystem can...
- C Chemistry . What is proper formula for 2 atoms of lithium for every atom of oxygen?...

Ответ:
4.33g
Explanation:
The reaction equation is given as:
2HgO → 2Hg + O₂
Given parameters:
Mass of HgO = 58.67g
Mass of Hg produced = 54.34g
Unknown:
Mass of oxygen produced = ?
Solution
From the reaction equation, using the mole concept, it is possible to determine the mass of oxygen gas produced.
To do this, we first determine the molar composition of the mass of the given mercury oxide. Then we use the balanced reaction equation to determine the mass of oxygen produced:
Number of moles of HgO =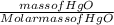
Molar mass of HgO = 200.6 + 16 = 216.6gmol⁻¹
Number of moles of HgO =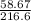 = 0.271mol
= 0.271mol
From the reaction equation, we know that:
2moles of HgO produced 1 mole of O₂
0.271moles of HgO would also produce, 0.136mol
Therefore, mass of O₂ gas produced = number of moles of O₂ x molar mass
Molar mass of O₂ = 16x2 = 32gmol⁻¹
Mass of O₂ gas = 0.136 x 32 = 4.33g
Ответ:
moles of Fe₂O₃ = mass ÷ molar mass
= 100 g ÷ [(56 × 2) + (16 × 2)] g/mol
= 100 g ÷ 144 g/mol
= 0.694 mol
mole ratio of Fe₂O₃ : CO is 1 : 3
∴ if mole of Fe₂O₃ = 0.694 mol
then mole of CO = 0.694 × 3
= 2.083 mol
mass of CO = mole × molar mass
= 2.083 × (12 + 16) g/mol
≈ 58.33 g