If energy is added to a boiling liquid what happens to the temperature of the liquid
Solved
Show answers
More tips
- A Art and Culture The History and Characteristics of Jazz Bands: A Deep Dive...
- W Work and Career Can Skill Alone Make You a Professional?...
- F Family and Home Daughter says: If you don t want to do it, don t do it. Should we persuade her?...
- S Science and Technology How to Secure Exam Sessions: Silence Mobile Phones in the Classroom...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- P Philosophy Is Everything We Strive for Eventually Achieved and Destroyed?...
- S Society and Politics Understanding Politics and Its Role in the Development of Civilization...
- P Philosophy Why Did God Create Man and Place Him in Obscurity?...
- S Society and Politics Skoptsy: Who They Are and How They Perform Castration?...
- O Other Childhood Fears: What Many of Us Experienced...
Answers on questions: Chemistry
- C Chemistry Why is the use of high-frequency radio waves a beneficial technological advancement for humans? Explain. A. They have the best penetration. B. They carry more information....
- E Engineering The surface of a 15-cm-diameter sphere is maintained at a temperature of 85°c. if the sphere is enclosed in a large room at 20°c, determine the heat transfer from the...
- M Mathematics Find the discriminant of 3p^2-6p+8=0 and give the number and type of solutions to the equation...
- E English Which word from this passage of matthew arnold s dover beach gives the poem a gloomy mood? a. calm b. gleams c. moon-blanched d. full...
- P Physics Which type of intermolecular attractive force operates between all molecules?...

Ответ:
1)Thus based on the diagram , the values of principle quantum number that can be assigned to the electrons are : 2,3 and 4.
2)This quantum number tells us the shell in which an electron is present and how far it is roughly from the nucleus.
Explanation:
According to the rules of electron filling in subshells in quantum mechanics , Aufbau's principle states that :
"An electron must be filled in a lower energy state or subshell and then higher. If they have the same energy , the one with lesser principal quantum number will be filled first."
Thus ,
The series of filling goes like:
Thus , the subshells in the given information have 1,3 and 1 orbitals respectively. Thus it has to be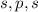 which is possible in two cases :
which is possible in two cases :
1)Thus based on the diagram , the values of principle quantum number that can be assigned to the electrons are : 2,3 and 4.
2)This quantum number tells us the shell in which an electron is present and how far it is roughly from the nucleus.