IsaacRodriguez245
06.08.2019 •
Chemistry
If you combine 27.1 g of a solute that has a molar mass of 27.1 g/mol with 100.0 g of a solvent, what is the molality of the resulting solution?
Solved
Show answers
More tips
- F Food and Cooking How to Quickly Put your Child to Sleep?...
- F Food and Cooking Discover the Benefits and Properties of Dates...
- C Computers and Internet Dynamically Assigned IP Address: What Is It and How Does It Work?...
- S Style and Beauty How to Get Rid of Acne: Scientifically Proven Methods...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
Answers on questions: Chemistry
- C Chemistry Use to view the image and contains a lens with 10x magnification...
- C Chemistry Which type of energy is stored in the foods that you eat? URGENT NEED ANSWER...
- C Chemistry A gas mixture is composed of 4.3 grams of hydrogen (density at STP= 0.089g/L) and 2.9grams of helium ( density at STP = 0.179 g/L). What is the percentage (v/v) helium...
- C Chemistry Which description correctly identifies the cation, the anion, and the formula for the compound they create? a cation = cl– anion = h+ formula = hcl b cation = mg+ anion...
- C Chemistry Write and balance chemical equations for gas producing reaction with sodium carbonate...
- C Chemistry Compare the benefits and disadvantages to the different alternative energy technologies....
- C Chemistry For every liter of sea water that evaporates, 3.7 g of magnesium hydroxide are produced. how many liters of sea water must evaporate to produce 5.00 moles of magnesium...
- C Chemistry Latoya performs an experiment using the lab equipment shown. which two properties is latoya most likely measuring?...
- C Chemistry When a reader makes inferences based on the details provided, it enables the reader to...
- C Chemistry Name three ways that scientists can ensure their results are reliable.Name three ways that scientists can ensure their results are reliable....

Ответ:
The molality of the solution is 0.1 m.
Explanation:
To calculate the molality of solution, we use the equation:
Where,
Putting values in above equation, we get:
Hence, the molality of the solution is 0.1 m.
Ответ:
the molality of the solution is 10 mol/kg
Explanation:
To find the molality of a solution we apply the following formula
data:
100 g of solvente
We have the amount of solvent expressed in grams but to be able to use the data in the formula we must pass it to kilograms
we apply a simple rule of three
27.1g of solute, molar mass 27.1 g/mol
We need the moles of solute and they give us grams of solute and molar mass
The molar mass is 27.1 g / mol This means that there is 27.1 g for each mol of solute.
The amount of solute we have is 27.1g and according to the molar mass this amount of solute corresponds exactly to 1 mole
Therefore having 27.1 g of solute is equivalent to saying that we have 1 mol of solute
Now that we have the data in the correct units we apply the formula
the molality of the solution is 10 mol/kg
Ответ:
Answer : The weight of water is 9 g
Explanation:
According to avogadro's law, 1 mole of every substance weighs equal to its molecular mass and contains avogadro's number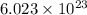 of particles.
of particles.
To calculate the number of moles, we use the equation:
1 mole of water weighs = 18 g
weighs = 18 g
0.5 moles of water weighs =
weighs = 
Thus the weight of water is 9 g