raymond5799
17.09.2019 •
Chemistry
If you were trying to heat soup, would you want to use a good thermal
conductor or a good thermal insulator? explain your answer.
Solved
Show answers
More tips
- D Dating, Love, Relationships Why do we feel shame?...
- S Style and Beauty How to Properly Apply Eye Makeup: Tips from a Professional Makeup Artist...
- A Animals and plants How ants survive winter: exploring the secrets of their winter life...
- F Food and Cooking Discover How to Properly Prepare Dough for Rasstegai...
- P Philosophy Unidentified Flying Object - What is the Nature of this Phenomenon?...
- F Family and Home Protect Your Home or Apartment from Pesky Ants...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
Answers on questions: Chemistry
- C Chemistry If the hydrogen ion concentration, [H+], in a solution is 2.65 x 10-2 M, what is [OH-]?...
- C Chemistry If the equilibrium concentration of O2 in air at 25∘C is 8×10−3 M, what is the equilibrium concentration of O3?...
- P Physics What are the forces that resist motion that make Newton s 1st Law hard to completely visualize on Earth?...
- E English Which of the following is a complex sentence? a. i simply cannot make heads or tails of rap lyrics, but i suppose i m just too old to understand them. b. as jake was reading...
- H Health Acommon workout myth is the more you the more calories you are burning. a) eat b) sweat c) watch tv d) play video games...
- M Mathematics Please answer this question...

Ответ:
Ответ:
pOH of resulting solution is 0.086
Explanation:
KOH and CsOH are monoacidic strong base
Number of moles of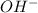 in 375 mL of 0.88 M of KOH =
in 375 mL of 0.88 M of KOH = 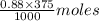 = 0.33 moles
= 0.33 moles
Number of moles of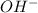 in 496 mL of 0.76 M of CsOH =
in 496 mL of 0.76 M of CsOH = 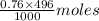 = 0.38 moles
= 0.38 moles
Total volume of mixture = (375 + 496) mL = 871 mL
Total number of moles of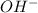 in mixture = (0.33 + 0.38) moles = 0.71 moles
in mixture = (0.33 + 0.38) moles = 0.71 moles
So, concentration of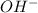 in mixture,
in mixture, ![[OH^{-}]](/tpl/images/0538/8908/e46dd.png) =
= 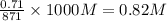
Hence,![pOH=-log[OH^{-}]=-log(0.82)=0.086](/tpl/images/0538/8908/1f1db.png)