rachelsweeney10
08.01.2020 •
Chemistry
Ihave no idea how to do this and it’s due tomorrow pls
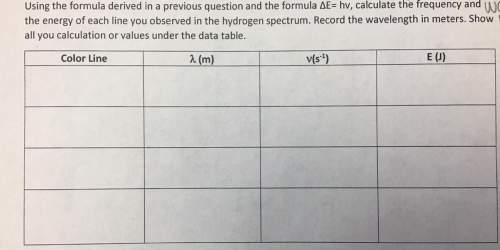
Solved
Show answers
More tips
- H Health and Medicine What to Do If You Are Bitten by a Dog?...
- A Animals and plants How ants survive winter: exploring the secrets of their winter life...
- F Food and Cooking Discover How to Properly Prepare Dough for Rasstegai...
- P Philosophy Unidentified Flying Object - What is the Nature of this Phenomenon?...
- F Family and Home Protect Your Home or Apartment from Pesky Ants...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
Answers on questions: Chemistry
- C Chemistry Which statement is false...
- C Chemistry Why atoms react and combine with others to form molecular compounds...
- C Chemistry Someone wanna talk with me bcs im so bored i can help with schoolwork to...
- C Chemistry A company spent the following on new product development last year: Product #1: $500,000 Product #2: $1,300,000 Product #3: $750,000 Product #4: $200,000 Product #5: $600,000 What...
- C Chemistry which of the following is an organism? a. water b. bacteria c. soil...
- C Chemistry If it takes 54ml of 0.1 m naoh to neutralize 125 ml of an hci solution,what is the concentration of the hci...
- C Chemistry What is the volume of 0.5 moles of gas at STP?...
- C Chemistry When your in a group online for your science class and your paired with two people who stutter smh...
- C Chemistry Question 2 (1 point) Which of the following does NOT involve a chemical reaction? Fallen leaves decay in a compost pile. Leaves change color in the fall. O A fire burns a pile of fallen...
- C Chemistry How many moles are in 84.23 g of ZnCl 2 ? Please help...

Ответ:
answer:
emission spectra are produced by thin gases in which the atoms do not experience many collisions (because of the low density). the emission lines correspond to photons of discrete energies that are emitted when excited atomic states in the gas make transitions back to lower-lying levels.
explanation: