In a living organism, the decay of c-14 produces 15.3 disintegrations per minute per gram of carbon. the half-life of c-14 is 5730 years. a bone sample with 2.8 g of carbon has 36.7 disintegrations per minute. how old is the bone sample in years?
Solved
Show answers
More tips
- F Food and Cooking Discovering the Benefits of Kiwi: From Its Nutritious Properties to Its Unusual Benefits...
- S Style and Beauty Discover the Art of Nail Design: How Do You Paint Your Nails?...
- P Philosophy How to Develop Extrasensory Abilities?...
- O Other Everything You Need to Know About Kudyabliks...
- C Computers and Internet The Twitter Phenomenon: What it is and How to Use it...
- C Computers and Internet How to Choose a Laptop: Expert Guide and Tips...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
Answers on questions: Chemistry
- M Mathematics The point slope formula (if possible) to write an equation of the line given the following information…we...
- A Advanced Placement (AP) what are the differences between relocation and expansion diffusion. explain and give a real world example of the different types and subtypes of diffusion using geographic...
- M Mathematics AB = Round your answer to the nearest hundredth. А ? 4 35° B С...
- H History Why do we have elections every r to 8 years and why are they important?...

Ответ:
The bone sample is 1278.86 years.
Explanation:
Using the integrated rate law:
N=
where:
k is the rate constant, N is the number of atoms at time t, No is the number of atoms at time t=0.
The rate constant can be determined using:
k = 0.693/
where is the half life which is equal to 5730 years
is the half life which is equal to 5730 years
Thus:
k = 0.693/5730 = 1.210* (1/year)
(1/year)
Then, we calculate the current rate. The organism produces 36.7 disintegrations per minute per 2.8 g. However, the rate constant is in years. Therefore, we convert the unit to disintegrations per year per gram.
To determine N, we use:
r=kN
where r is the current rate which is 6.724*
Thus:
N = 5.557*
Similarly, we have 15.3 disintegrations per minute per gram of carbon. Therefore,
To determine No, we use:
r=kNo
Thus:
No = 6.487*
Therefore using the integrated rate law:
N=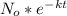
5.557*
0.8567 =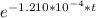
Taking the natural log (ln) of both sides which is the inverse of exponential, we have:
ln (0.8567) = ln (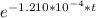
-0.1547 = -1.210*
t = 1278.86 years. Therefore, the bone sample is 1278.86 years old.
Ответ:
lol you funny
Explanation: