In a titration of 35.00 mL of 0.737 M H2SO4, mL of a 0.827 M KOH solution is required for neutralization.
A) 35.0
B) 1.12
C) 25.8
D) 62.4
E) 39.3
Solved
Show answers
More tips
- H Health and Medicine How to Calm Your Nerves? Expert Tips That Actually Work...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- A Animals and plants 5 Tips for Taking Care of Yews to Keep Them Green and Beautiful...
- S Sport How to wrap boxing hand wraps? Everything you need to know!...
- F Food and Cooking 10 Reasons Why You Should Avoid Giving Re-Gifts: An Informative Guide...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Style and Beauty How Are Eyelash Extensions Applied? All Your Questions Answered...
- F Food and Cooking 10 Tips for Proper Sushi Consumption...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
Answers on questions: Chemistry
- C Chemistry What will happen wen lead react with copper 2 sulfate...
- C Chemistry The number marked in red in front of an ammonia 4 NH3 molecule is called I. All choices are correct II. Coefficient III. - shows how many atoms of an element are in a molecule...
- C Chemistry Can someone please help????...
- C Chemistry 10) Which gas sample at STP has the same number of molecules as a 2.0 Liter sample of Cl2 (g) at STP? a) 1.0 L of H2 (g) b) 2.0 L of He c) 3.0 L of NH3 d) 4.0 L of O2 (g)...
- C Chemistry Vice Chancellor of ABUAD...
- C Chemistry How many moles are 9.61 x 1021 atoms of iron?(show work)...
- C Chemistry I meant to put Physics Please Help Me I need help i ask this question yesterday and someone deleted it please ill mark brainliest the question is in the pictures thank...
- C Chemistry How many molecules are there in 20.0L of O2 at stp...
- M Mathematics Fernanda bought a 500-milliliter container juice. she used some of the juice and has 200 milliliters left. how much orange juice did she use?...
- H History You can t keep me out. all of the following were contributing factors to the fall of the persian empire a. constant plots to take over the throne c. defeat of persian army...

Ответ:
In a titration of 35.00 mL of 0.737 M H₂SO₄, 62.4 mL of a 0.827 M KOH solution is required for neutralization.
Explanation:
The balanced reaction is
H₂SO₄ + 2 KOH ⇒ 2 H₂O + K₂SO₄
By stoichiometry of the reaction (that is, the relationship between the amount of reagents and products in a chemical reaction) 1 mole of H₂SO₄ is neutralized with 2 moles of KOH.
The molarity M being the number of moles of solute that are dissolved in a given volume, expressed as:
in units of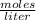
then the number of moles can be calculated as:
number of moles= molarity* volume
You have acid H₂SO₄
35.00 mL= 0.035 L (being 1,000 mL= 1 L) Molarity= 0.737 MThen:
number of moles= 0.737 M* 0.035 L
number of moles= 0.0258
So you must neutralize 0.0258 moles of H₂SO₄. Now you can apply the following rule of three: if by stoichiometry 1 mole of H₂SO₄ are neutralized with 2 moles of KOH, 0.0258 moles of H₂SO₄ are neutralized with how many moles of KOH?
moles of KOH= 0.0516
Then 0.0516 moles of KOH are needed. So you know:
Molarity= 0.827 Mnumber of moles= 0.0516volume=?Replacing in the definition of molarity:
Solving:
volume=0.0624 L= 62.4 mL
In a titration of 35.00 mL of 0.737 M H₂SO₄, 62.4 mL of a 0.827 M KOH solution is required for neutralization.
Ответ:
Explanation:
1 Toi et ton frere vous ete tres grand
2 Moi je suis grand et blond
3 Emma est mince et migone
4 Comment est le professeur d'anglais
5 Ils ne sont pas gros
6 Alicia et Jeanne sont gentillent et intelligentes
7 Eva est marrante mais timide
8 J'ai trois frere penibles mais beau