TerronRice
12.01.2021 •
Chemistry
In the reaction Zn + CuSO₄ → Cu + ZnSO₄
Solved
Show answers
More tips
- D Dating, Love, Relationships How to Overcome Jealousy: Tips and Tricks...
- H Health and Medicine Headache: A Comprehensive Guide to Treatment...
- G Goods and services Stock center - a modern way of optimizing logistics...
- F Food and Cooking How to Properly Wash a Down Jacket? Tips from Experts...
- C Computers and Internet Thin Client: What It Is and Why You Need It?...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- C Computers and Internet Clearing Cache: How to Speed Up Your Browser...
- S Style and Beauty How are artificial nails removed?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
Answers on questions: Chemistry
- C Chemistry Carbonic anhydrase is a zinc-based enzyme that catalyzes the conversion of carbon dioxide to carbonic acid. in an experiment to study its eff ect, it was found that the...
- C Chemistry Agas occupies 3.5 l at standard pressure. find the volume of the gas when the pressure is 1140 mm hg....
- C Chemistry Which of the following is NOT. A property of salt in the ionic and covalent properties lab?...
- C Chemistry 3) _Na + |H,80_ - _Na,304 _Na2SO4 + _H2...
- C Chemistry A student moves the end of a compressed coiled spring up and down to demonstrate wave motion she then moves the compressed coiled spring faster but keeps everything else...
- M Medicine QUESTION 19 Which of the following is a technological advancement for treatment of certain medical conditions...
- C Computers and Technology Which of the following is an example of a distant social network? a.One s immediate family members b.Members of a street gang c.One s girlfriend or boyfriend d.LinkedIn...
- B Business Last year Tiemann Technologies reported $10,500 of sales, $6,250 of operating costs other than depreciation, and $1,300 of depreciation. The company had no amortization...
- S Social Studies Imagine that you are in Hong Kong reading the morning news and you notice a headline about a double murder that took place overnight. A suspect is in custody. Which of...
- E English Attitudes do a good job predicting behavior when: There is discord among the affective and cognitive components of the attitude. Attitudes and behavior are measured at...

Ответ:
The given question is incomplete. The complete question is:
In the reaction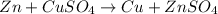 , identify the oxidizing and reducing agent.
, identify the oxidizing and reducing agent.
reducing agent. : Zn
oxidizing agent :
Explanation:
Oxidation reaction : When there is an increase in oxidation state number.
Reduction reaction : when there is a decrease in oxidation state number.
Zinc metal has undergone oxidation, as its oxidation state is changing from 0 to 2+. Zinc metal is getting converted into zinc ions.
The chemical agent which itself get oxidized and reduce others is called reducing agent.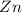 is a reducing agent.
is a reducing agent.
The chemical agent which itself get reduced and oxidize others is called oxidizing agent. is an oxidizing agent.
is an oxidizing agent.
Ответ:
a) 2
b) 1/4
c) 9
Explanation:
a) for a first order reaction in reactant A
r initial = k*[A initial]
then if the concentration is doubled [A final ]= 2*[A initial] , then
r final = k*[A final ] = 2* k*[A initial] = 2*r initial
then the velocity changes by a factor of 2
b) for a second order reaction in reactant B
r initial = k*[B initial]²
then if the concentration is halved: [B final ]= [B initial]/2 , then
r final = k*[B final ]² = k*( [B initial]/2 )² =k* [B initial]² /4 = r initial /4
then the velocity changes by a factor of 1/4
c) for a second order reaction in reactant C
r initial = k*[C initial]²
then if the concentration is tripled : [C final ]= 3* [C initial] , then
r final = k*[C final ]² = k*( 3*[C initial] )² =k* [C initial]² *9 = 9 * r initial
then the velocity changes by a factor of 9