Iron (iii) chloride + sodium carbonate → iron (iii) carbonate + sodium chloride which of these is the balanced equation for the reaction described above?
Solved
Show answers
More tips
- L Leisure and Entertainment How to Choose a Program for Cutting Music?...
- A Auto and Moto How to choose the right drive for your BMW...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
Answers on questions: Chemistry
- C Chemistry What is the wavelength of electromagnetic radiation with a frequency of 5.00 x 10^12 Hz? S radio infrared ultraviolet gamma rays...
- M Mathematics What is the domain of the function graphed below?...
- M Mathematics 20 points find the area under one arch of a cycloid described by the parametric equations x = 3(20 - sin 20) and y = 3( i - cos 20). use 0 and π for the limiting values of θ ....
- M Mathematics Amos is a waiter. He earns $3.50 per hour plus tips. Last weekend, Amos earned $150.00 in tips and more than $206.00 total. Write an inequality for this situation where h represents...

Ответ:
Ответ:
Answer : The percentage by mass of carbon in hydrocortisone valerate is 69.95 %.
Solution :
The molecular formula is .
.
The molar mass of Carbon = 12 g/mole
The molar mass of Hydrogen = 1 g/mole
The molar mass of Oxygen = 16 g/mole
First we have to calculate the mass of one mole of .
.
In this molecule, there are 26 carbon atom, 38 hydrogen atom and 6 oxygen atom.
Mass of 26 moles of carbon = 26 moles × 12 g/mole = 312 g
Mass of 38 moles of hydrogen = 38 moles × 1 g/mole = 38 g
Mass of 6 moles of oxygen = 6 moles × 16 g/mole = 96 g
Total mass of one mole of = 312 + 38 + 96 = 446 g
= 312 + 38 + 96 = 446 g
Now we have to calculate the percent by mass of carbon in hydrocortisone valerate.
%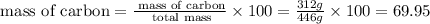 %
%
Therefore, the percentage by mass of carbon in hydrocortisone valerate is 69.95 %.