davelopez979
26.07.2021 •
Chemistry
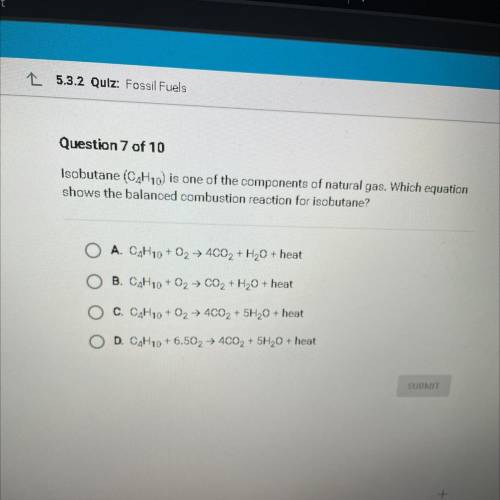
Isobutane (C4H10) is one of the components of natural gas. Which equation
shows the balanced combustion reaction for isobutane?
A. C4H10 + O2 + 4CO2 + H2O + heat
B. C4H10 + O2 + CO2 + H2O + heat
C. C4H10 + O2 + 4C02 + 5H20 + heat
D. C4H10 +6.502 → 4002 + 5H20 + heat
SUBMIT
Solved
Show answers
More tips
- G Goods and services How to Choose the Best Publishing House for Your Children s Book: Tips and Recommendations...
- S Science and Technology When do we change our clocks?...
- A Animals and plants How to Teach Your Parrot to Talk?...
- F Family and Home How to Remove Fading from Clothes: Tips and Tricks...
- F Food and Cooking How to Make Polendwitsa at Home?...
- F Family and Home Parents or Environment: Who Has the Most Influence on a Child s Upbringing?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
Answers on questions: Chemistry
- C Chemistry Each electron has a charge of 1–, and each proton has a charge of 1+. You can calculate the charge of an atom by subtracting the number of electrons from the number of...
- S Social Studies In Florida, when spring changes to summer, what seasonal changes do plants experience? (1 point) a Plants begin to change color and lose their leaves before they can germinate....
- P Physics Acart of mass 250 g is place on a frictionless horizontal air track. a spring having a spring constant of 9.5 n/m is attached between the cart and the left end o f the...
- E English Dear Tony, Colleen’s wedding will begin at noon at 455 W. Chestnut Drive. There will be a reception after the ceremony, and of course everyone is invited. It is going to...
- M Mathematics A cooking competition rated each participant s dish on both appearance and taste according to a 10 point scale. An approximate least-squares regression line was used to...

Ответ:
D. C₄H₁₀ + 6.5O₂ → 4CO₂ + 5H₂O + heat
Explanation:
The reaction of the combustion of isobutane with oxygen is presented as follows;
C₄H₁₀ + 6.5O₂ → 4CO₂ + 5H₂O + heat
In the above reaction, the number of elements on the reactant side are;
4 carbon, C, atoms, 10 hydrogen, H, atoms, and 13 oxygen, O, atoms
The number of elements on the product side of the above chemical reaction are;
The number of carbon atoms, C = 4
The number of hydrogen atoms, H = 10
The number of oxygen atoms, O = 13
Therefore, the number of atoms on the reactant side of the chemical equation are equal to the number of atoms on the product side, and the combustion reaction for isobutane is balanced.
Ответ: