It's not unusual for students who see this demonstration to claim that the "red stuff" is rust (meaning the compound iron III oxide). Brainstorm some empirical data (information that you could collect in the lab) that would prove or disprove the claim that the "red stuff" is rust.
Solved
Show answers
More tips
Answers on questions: Chemistry
- C Chemistry What is the iupac name for the compound zno? (1) zinc oxide (2) zinc oxalate(3) zinc peroxide (4) zinc hydroxide...
- M Mathematics A warren of rabbits has a population of 350. The population is increasing at a rate of 3% per year. How can you write an exponential growth function to find the...
- P Physics Question 2 A) A spring is compressed, resulting in its displacement to the right. What happens to the spring when it is released? (1 point) The spring exerts a...
- M Mathematics Step-by-step explanation: If you use the formula P(r)=nCrP^rq^n-r. P is the probability of success = .25 n is the total number of questions = 8 r is the total number...
- C Chemistry Is the concentration of 25 cm3 solution same as 250 cm3 solution?...
- C Chemistry Rubidium will react with oxygen in which of the following formulas? RbO RbO2 Rb2O Rb3O2...

Ответ:
Rust is muddy red in color.
Explanation:
When iron or iron alloys are exposed to atmospheric air which contains moisture, it gets oxidized and forms rust. Rust is not desirable. The piece of iron converts into iron oxide when it is exposed to moisture or oxygen due to oxidation reaction.
Rust gives a flaky reddish brown color to the metal and it becomes more worse over the time. Rust consists of hydrated iron (3) oxides i.e. and iron(III) oxide-hydroxide i.e.
and iron(III) oxide-hydroxide i.e. 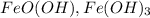 .
.
Ответ: