It takes 167 s for an unknown gas to effuse through a porous wall and 99 s for the same volume of n2 gas to effuse at the same temperature and pressure. what is the molar mass of the unknown gas?
Solved
Show answers
More tips
- B Business and Finance What is the Difference Between Visa and Visa Gold?...
- A Animals and plants Why do cats go crazy over catnip?...
- W Work and Career Everything You Need to Know About MBA Programs...
- S Sport How to Do Push-ups Correctly?...
- S Style and Beauty How to Grow Hair Faster: Real Methods and Advice...
- F Family and Home How to Remove Fading from Clothes: Tips and Tricks...
- F Food and Cooking How to Make Polendwitsa at Home?...
- F Family and Home Parents or Environment: Who Has the Most Influence on a Child s Upbringing?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
Answers on questions: Chemistry
- C Chemistry Name the following alkane molecule: A. methylpentane B. 2-methylpentane C. 2-ethylpentane...
- C Chemistry Name the following alkane molecule: CH3 CH3CCH3 CH3 A. 2-ethylpropane B. 2-dimethylpropane C. 2,2-dimethylpropane...
- C Chemistry Why is the r.a.m. Value used?...
- C Chemistry Place the letters on the spectrum below from least scientific two more scientific A. All living things are composed of one or more cells. We know this because every...
- C Chemistry If particles of a solid move slow and particles of a gas move fast, which would have higher kinetic energy (KE)?...
- C Chemistry Anu-hmkv-jzo all gi.rls are welcomed to see my cream...
- C Chemistry Which of the following most likely happens during a chemical change? a.no new substances are formed. b.properties of a substance remain unchanged. c.composition of a...
- C Chemistry How do you calculate the pressure when given the volume, number of moles, and the temperature? (Step by step example please and thank you)...
- C Chemistry Dar un ejemplo de reacciones anaerobicas (tema : reacciones quimicas)...
- C Chemistry Descriptive Lab Report Guide exploring cells answer...

Ответ:
Answer : The molar mass of the unknown gas will be 79.7 g/mol
Explanation : To solve this question we can use graham's law;
Now we can use nitrogen as the gas number 2, which travels faster than gas 1;
So, 167 / 99 = 1.687 So the nitrogen gas is 1.687 times faster that the unknown gas 1
We can compare the rates of both the gases;
So here, Rate of gas 2 / Rate of gas 1 =
Now, 1.687 = square root [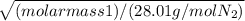 ]
]
When we square both the sides we get;
2.845 = (molar mass 1) / (28.01 g/mol N2)
On rearranging, we get,
2.845 X (28.01 g/mol N2) = Molar mass 1
So the molar mass of unknown gas will be = 79.7 g/mol
Ответ:
Since glass is translucent meaning you can see through it, heat energy from the sun will easily pass through it. If you were to conduct this test with a piece of metal the heat would be absorbed by the metal making it extremely hot. In this instance when they put a piece of cardboard in front of the glass it would absorb the heat energy since it is not translucent.