naisayaperry
25.06.2021 •
Chemistry
IUPAC name of this compound??
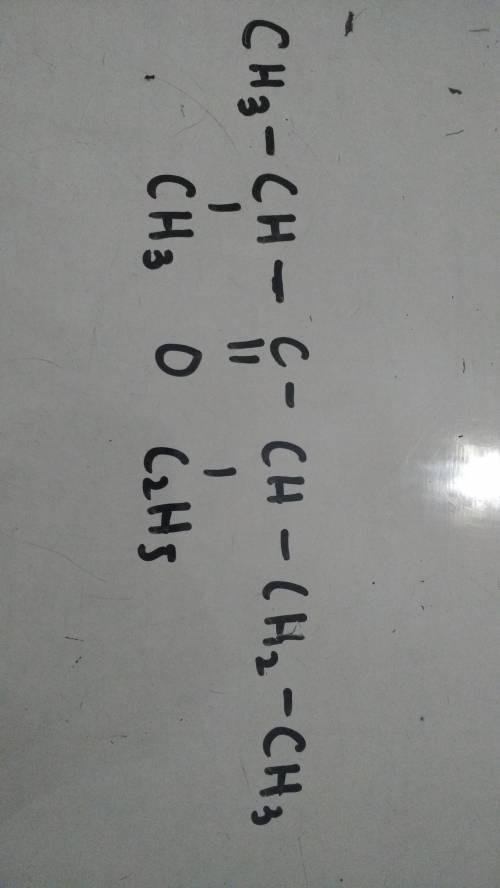
Solved
Show answers
More tips
- F Food and Cooking Learn How to Make Ice Cream at Home - Step by Step Guide...
- H Health and Medicine When can it be said that a person has a normal pulse?...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- L Leisure and Entertainment Carving: History and Techniques for Creating Vegetable and Fruit Decorations...
Answers on questions: Chemistry
- C Chemistry What chemical property does a wick have that wax doesn’t...
- C Chemistry What is the electron configuration for Sulfur?...
- C Chemistry Acombustion analysis of a 0.503 g sample of an unknown hydrocarbon yields 1.53 g co 2 2 and 0.756 g h 2 2 o. what is the empirical formula of the sample? give your answer...
- C Chemistry Which organism would most likely be the main source of oxygen in a forest ecosystem?...
- C Chemistry A set of 13 particles occupies states with energies of 0, 100, and 200 cm^-1. Calculate the total energy and number of microstates for the following energy configurations....
- M Mathematics Find the set of maximum and minimum values of the following function: y=3sinx+4cosx+1...
- M Mathematics What is 1000000000*20009643...
- M Mathematics Find the value of 17 - a when a = 7. X 3 ?...
- B Business Roy and Brandi are engaged and plan to get married. Roy is a full-time student and earns $9,000 from a part-time job. With this income, student loans, savings, and nontaxable...
- M Mathematics They dont help me anymoreeeeeeeeeeee...

Ответ:
4-ethyl 2-methylhexan-3-one is the compound in the above picture
Ответ:
λ(Decay constant) = 0.0492sec-¹
The relationship between Decay Constant and Half life (t½)
λ=0.693/t½
given the decay constant as 0.0492
We can get the Half life
t½ = 0.693/0.0492
t½= 14.09secs.
N = N°(½)ⁿ
Where N= Remaining Amount of the original Compound
N° = Original Amount of compound.
n is simply a tern used to represent the ratio of time taken/half life
In other words
n=t/t½
Given N°=0.656 mol/L
N=0.433mol/L
0.433 = 0.656(½)ⁿ
(½)ⁿ = 0.433/0.656
(½)ⁿ = 0.660
Taking the Natural Log(Ln) of both sides
Ln(½)ⁿ = Ln(0.66)
nLn(½) = -0.4155
n(-0.6931) = -0.4155
n= 0.4155/0.6931
n= 0.5995.
n = time/half life
t = half life x n
t = 14.09 x 0.5995
t = 8.45secs.