jessnolonger
25.06.2019 •
Chemistry
Jake collected samples of two substances while he was out walking. after taking the samples home, he ran tests and found that one substance is slippery and conducts electricity in water. these properties made jake conclude that the substance is probably . the other substance continuously made bubbles of hydrogen gas when jake dropped magnesium into an aqueous solution of the substance. jake concluded that the second substance is probably .
Solved
Show answers
More tips
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- F Family and Home How to Remove Tar Stains: Tips and Recommendations from Experts...
- F Family and Home How to Remove Fading from Clothes: Tips and Tricks...
- S Sport How to Do a Jumping Split...
- H Health and Medicine How Did Inna Lose Weight on Dom 2?...
- F Family and Home How to Properly Fold Napkins in a Napkin Holder?...
- F Food and Cooking How to Set Up Ventrilo - The Ultimate Guide...
- S Science and Technology How to Make a Homemade Smoker: The Ultimate Guide...
- A Auto and Moto Battle for the Relocation of The Cherkizovsky Market: Who Won?...
- C Computers and Internet How Do You Refill Cartridges?...
Answers on questions: Chemistry
- C Chemistry Which two chemical equations model double-replacement reactions? A. 2Na + MgCl2 + 2NaCl + Mg - B. AgNO3 + Lici AGCI + LINO3 C. CH4 + 202 – 2H2O + CO2 OD. HC2H202 +...
- C Chemistry HELP! IM GIVING BRAINLIST.What is salinity? Question 1 options: The concentration of dissolved salt in a liquid to become incorporated with a liquid in forming a solution...
- C Chemistry Consider the reaction: m + 2hcl → mcl2 + h2 when 0.25 mol of the metal, m, reacted with an aqueous hcl solution (the hcl is in excess), the temperature of the solution...
- C Chemistry Hydrogen can be extracted from natural gas according to the following equilibrium. ch4(g) + co2(g) 2co(g) + 2h2(g) a reaction mixture consisting of 24.0 g of ch4 and...
- C Chemistry S8 + 4CL2 → 4S2Cl2 If 200.0 g of sulfur reacts with 100.0 g of chlorine according to this reaction, what mass of disulfur dichloride is produced? A 1.4 g of S2Cl2...
- C Chemistry Help needed ASAP, I will mark as...
- C Chemistry Help needed asap, will mark as...
- G Geography I like jeff hardy and why...
- S Social Studies How can exercising our imagination help us to develop a growth mindset...
- P Physics How much energy would be released if you converted 1.2 kg of matter entirely into energy? A. 1.3 x 10^17 J B. 1.2 x 10^34J C. 3.6 x 10^8J D. 1.1 x 10^17J...

Ответ:
The substance which is slippery and conducts electricity in water is base.
The substance which continuously made bubbles of hydrogen gas when Jake dropped magnesium into an aqueous solution of the substance is acid.
Explanation:
1. An acid is defined as a substance which dissociates to give ions when dissolved in water.
ions when dissolved in water.
They conduct electricity when dissolved in water due to the presence of ions.
Acids are sour in taste.
Acids react with metals to give hydrogen gas.
When magnesium is treated with a strong acid such as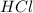 ,magnesium being more reactive than hydrogen displaces hydrogen from its salt solution and thus produce magnesium chloride and hydrogen gas.
,magnesium being more reactive than hydrogen displaces hydrogen from its salt solution and thus produce magnesium chloride and hydrogen gas.
2. A base is defined as a substance which dissociates to give when dissolved in water.
when dissolved in water.
They conduct electricity when dissolved in water due to the presence of ions.
Bases are slippery in nature.
Thus we can conclude that the substance which is slippery and conducts electricity in water is base and the substance which continuously made bubbles of hydrogen gas when Jake dropped magnesium into an aqueous solution of the substance is acid.
Ответ: