monkemily1
23.03.2020 •
Chemistry
Linolenic acid (C18H30O2 - M.W. = 278.42 g/mol) reacts with hydrogen gas according to the equation: C18H30O2 + 3H2 (g) → C18H36O2 What volume of hydrogen gas, as measured at P = 1.00 atm and T = 273.15 K, is required to react with 10.5 g of linolenic acid in this reaction?
Solved
Show answers
More tips
Answers on questions: Chemistry
- C Chemistry Which of the following determines the physical and chemical properties of a substance? [mark all correct answers] a. Size b. Atomic Structure c. Amount of Matter O d. Molecular...
- C Chemistry An object has a mass of 22.5 grams and a volume of 13.7 cm3. What is the density?...
- C Chemistry A student performed an investigation to see what would happen when an iron nail is added to a copper sulfate solution. Below is the chemical equation that shows what happens when...
- C Chemistry PLEASE HELP The pharynx is also known as the a. throat. b. cilia. c. bronchi. d. vocal cord. Please select the best answer from the choices provided...
- C Chemistry Scientists use what properties to them identify and classify matter...
- C Chemistry An generates heat for an entire building from one central source....
- C Chemistry on the planet mars the average night temperature is 24k. what is the temperature in degrees celsius and degree fahrehheit...
- C Chemistry Describe how thermal shock can break an ordinary sheet of glass...
- C Chemistry Most enzymes in living things are made up of a. carbohydrates b. lipids c. proteins d. nucleic acids...
- P Physics How many inches is equivalent to 4.02 km? ( 1 mi = 1609 m ) ( 5280 ft = 1 mi ) show your work ! : )...

Ответ:
2.53 L is the volume of H₂ needed
Explanation:
The reaction is: C₁₈H₃₀O₂ + 3H₂ → C₁₈H₃₆O₂
By the way we can say, that 1 mol of linolenic acid reacts with 3 moles of oxygen in order to produce, 1 mol of stearic acid.
By stoichiometry, ratio is 1:3
Let's convert the mass of the linolenic acid to moles:
10.5 g . 1 mol / 278.42 g = 0.0377 moles
We apply a rule of three:
1 mol of linolenic acid needs 3 moles of H₂ to react
Then, 0.0377 moles will react with (0.0377 . 3 )/1 = 0.113 moles of hydrogen
We apply the Ideal Gases Law to find out the volume (condition of measure are STP) → P . V = n . R . T → V = ( n . R .T ) / P
V = (0.113 mol . 0.082 L.atm/mol.K . 273.15K) 1 atm = 2.53 L
Ответ:
ionization energy or ionization energy is the minimum amount of energy required to remove the valence electron i.e. the outmost orbital electron.
_____________________________________Magnesium Electronic Configuration: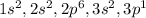
_____________________________________Question:Why does magnesium has more ionization energy than Aluminum?
Reason:Aluminum's has one unpaired electron in it's Valence energy orbital i.e. 3p. the electrons are not paired that means it has only electron in the last orbital that's why it is written as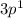 , and Magnesium's Valence Energy Orbital i.e. 3s. the electrons are paired that means it has 2 electron that's why it is written as
, and Magnesium's Valence Energy Orbital i.e. 3s. the electrons are paired that means it has 2 electron that's why it is written as 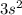 . It is energetically favorable for all the electrons in an orbital to be paired, which means that breaking up this pair would require more energy, that is why magnesium has more ionization energy. The Aluminum does not have paired electrons thus they are removed much easily than the paired electrons in the Magnesium.
. It is energetically favorable for all the electrons in an orbital to be paired, which means that breaking up this pair would require more energy, that is why magnesium has more ionization energy. The Aluminum does not have paired electrons thus they are removed much easily than the paired electrons in the Magnesium.
_____________________________________Best Regards,'Borz'