List the following solutions in order of decreasing freezing point: I. 0.040 mol kg−1 glycerin (a nonelectrolyte) II. 0.025 mol kg−1 Na2SO4 (assume complete dissociation in aqueous solution) III. 0.030 mol kg−1 KBr (assume complete dissociation in aqueous solution)
Solved
Show answers
More tips
- H Health and Medicine When can it be said that a person has a normal pulse?...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- L Leisure and Entertainment Carving: History and Techniques for Creating Vegetable and Fruit Decorations...
- P Photography and Videography How to Choose the Perfect Photo Paper for Your Images?...

Ответ:
0.040 mol glycerin > 0.030 mol KBr> 0.025 mol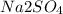
Explanation:
Depression in freezing point:
where,
m = molality
1. For 0.040 mol glycerin
, i= 1 as it is a non electrolyte and does not dissociate, concentration of ions will be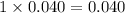
2. For 0.025 mol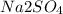
, i= 3 as it is a electrolyte and dissociate to give 3 ions. and concentration of ions will be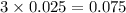
3. For 0.030 mol KBr
, i= 2 as it is a electrolyte and dissociate to give 2 ions, concentration of ions will be
Thus as concentration of solute is highest for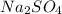 , the depression in freezing point will be highest and thus freezing point will be lowest.
, the depression in freezing point will be highest and thus freezing point will be lowest.
The order of decreasing freezing point: 0.040 mol glycerin > 0.030 mol KBr> 0.025 mol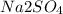
Ответ:
Type of Chemical Reaction: For this reaction we have a single displacement reaction. Balancing Strategies: Note that this is the chemical reaction that takes place when diluted Sulfuric acid reacts with iron.
Explanation: