Magnesium metal with the electron configuration [Ne] 3S2 bonds with the nonmetal Fluorine with the electron configuration [He] 2s2 2p5. The correct chemical formula is A) Mg2F B) Mg2F7 C) MgF D) MgF2
Solved
Show answers
More tips
- O Other How to Accidentally Get a Rare Coin with Your Change and How to Know Its Value?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- D Dating, Love, Relationships Why do we feel shame?...
- F Family and Home Why Does God Punish Us Constantly and How Can We Fix It?...
- F Family and Home Do Lullabies Help Babies Sleep or Is it Just a Myth?...
- B Business and Finance How to Get Your Money Back When Lending? Top 7 Ways...
- S Society and Politics Выборы: Смысл, Значение и Отражение...
- D Dating, Love, Relationships Why Should the Man be Active and the Woman Passive during Foreplay?...
- P Philosophy How did the concept of module arise in computer science?...
- S Society and Politics Why are thugs called gopniks ? A fascinating journey through Russian subculture...
Answers on questions: Chemistry
- C Chemistry Please help me with this question....
- M Mathematics Leonard used 2/7 of his paycheck to pay his phone bi and put 1/3 of his paycheck in savings. based on the amount of money left, which of the following amounts he could spend...
- M Mathematics A scientist looks at bacterium and in a virus in a lab the bacterium has a diameter of 10-6 meters. The virus has a diameter of 10-7 meters...
- C Chemistry How many c atoms are in 2 mol of sucrose?...

Ответ:
a) CO₂: 21,9%; CO: 7,0%; CH₄: 18,2%; H₂: 0,8%; N₂: 52,1%
b) 24,09 g/mol
Explanation:
a) It is posible to obtain the composition of the gas mixture in weight% using molecular mass of each compound, thus:
12% CO₂×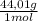 = 528,1 g
= 528,1 g
6% CO×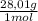 = 168,1 g
= 168,1 g
27,3% CH₄×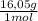 = 438,2 g
= 438,2 g
9,9% H₂×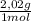 = 20,0 g
= 20,0 g
44,8% N₂×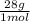 = 1254,4 g
= 1254,4 g
The total mass of the gas mixture is:
528,1g + 168,1g + 438,2g + 20,0g + 1254,4g = 2408,8 g
Thus composition of the gas mixture in weight% is:
CO₂: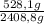 ×100 = 21,9%
×100 = 21,9%
CO: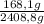 ×100 = 7,0%
×100 = 7,0%
CH₄: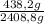 ×100 = 18,2%
×100 = 18,2%
H₂: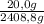 ×100 = 0,8%
×100 = 0,8%
N₂: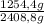 ×100 = 52,1%
×100 = 52,1%
b) The average molecular weight of the gas mixture is determined with mole % composition, thus:
0,12×44,01g/mol + 0,06×28,01g/mol + 0,273×16,05g/mol + 0,099×2,02g/mol + 0,448×28g/mol = 24,09 g/mol
I hope it helps!