ljcervantes4824
25.09.2019 •
Chemistry
Mol of an ideal gas expands through a turbine adiabatically to produce work. assuming steady flow conditions, how much work is lost due to the entropy generated, if the surroundings are at 300 k? give the value of lost work to the nearest). inlet conditions: 516.3 k and 6.3 bar outlet conditions: 386.2 k and 1.7 bar cp = (9/2)r where r = 8.314j/mol-k.
Solved
Show answers
More tips
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- L Leisure and Entertainment Carving: History and Techniques for Creating Vegetable and Fruit Decorations...
- P Photography and Videography How to Choose the Perfect Photo Paper for Your Images?...
- H Health and Medicine What vaccines do children need?...
- H Health and Medicine Reasons for the Appearance of Warts: Everything You Need to Know...
- A Art and Culture How to Learn Screaming: Step-by-Step Guide for Beginners...
- H Health and Medicine Contraceptive Pills After 35: The Importance Of Choosing The Right Medication...

Ответ:
Explanation:
According to avogadro's law, 1 mole of every substance occupies 22.4 Liters at STP and contains avogadro's number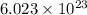 of particles.
of particles.
To calculate the moles, we use the equation:
Now 1 mole of molecule contains =
molecule contains = 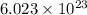 of molecules.
of molecules.
0.0016 moles of molecule contains =
molecule contains =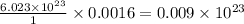 molecules.
molecules.